All published articles of this journal are available on ScienceDirect.
Effect of Short-Term Exposure to High Particulate Levels on Cough Reflex Sensitivity in Healthy Tourists: A Pilot Study
Abstract
Background:
Previous studies have reported a relationship between particulate air pollution and respiratory symptoms or decline in lung function, but information about acute effects of short-term exposure to airborne particulate matter (PM) on cough and pulmonary function is scarce.
Objective:
To investigate the effect of short-term exposure to high concentrations of PM on the cough reflex threshold, urge-to-cough, pulmonary function, and cough-related quality of life in a group of healthy non-resident volunteers visiting Beijing, China.
Methods:
Seventeen healthy residents of Sendai, Japan, who planned to attend a meeting in Beijing, were recruited. We checked local air quality and measured cough reflex thresholds, urge-to-cough, pulmonary function, and Leicester Cough Questionnaire-acute (LCQ-acute) scores in the volunteers before, during, and after their trip to Beijing.
Results:
The PM2.5 and PM10 concentrations in Beijing were significantly higher than those in Japan on the measurement days. Cough reflex thresholds, expressed as nebulized citric acid concentrations required to induce ≥ 2 and ≥ 5 coughs, were significantly lower during the stay in Beijing than before or after the visit. Vital capacity, forced expiratory volume in one second (FEV1), forced vital capacity (FVC), and FEV1/FVC were significantly lower during the stay in Beijing than before the trip. Similarly, the urge-to-cough threshold was significantly lower during the stay in Beijing than after the trip, as was the total LCQ-acute score.
Conclusion:
We tentatively concluded that short-term exposure to high PM concentrations may have adverse effects on cough reflex and urge-to-cough thresholds, pulmonary function, and cough-related quality of life.
INTRODUCTION
Several studies have reported that daily changes in the levels of ambient air pollution are associated with morbidity and mortality [1, 2]. Among the air pollutants, particulate matter (PM) has been associated with adverse health effects and therefore, extensively studied [3, 4]. The World Health Organization has set air quality guideline values for the daily mean concentrations of ambient PM2.5 (particle size smaller than 5 μm) at 25 μg/m3 and PM10 (particle size smaller than 10 μm) at 50 μg/m3. Epidemiological studies have shown associations between PM and respiratory symptoms including cough and phlegm [5-7]. Transient receptor potential (TRP)-class ankyrin-1 (TRPA1) and vanilloid-1 (TRPV1) ion channels are preferentially activated by environmental particulate matter, which is thought to contribute to the cough reflex [8-10]. Although previous reports support the hypothesis that coughing can be induced by exposure to environmental particulate matter, there are few reports on the effect of exposure to PM on cough reflex thresholds. Coughing is an important defensive reflex that removes foreign matter from the airways and there is a cognitive urge-to-cough sensory process that precedes the cough motor event [11]. In order to elucidate the association between ambient PM concentrations and cough, both cough reflex thresholds and urge-to-cough bear scrutiny.
Several laboratory-based studies have been used to examine the effects of exposure to airborne particulate matter on pulmonary function in humans [12, 13], but adequate research has not been carried out to measure acute changes in pulmonary function during exposure to actual day-to-day concentrations and durations of particulate air pollution.
A small group of people from Tohoku University in Sendai, Japan attended the Seventh World Congress of the International Society of Physical and Rehabilitation Medicine held at Beijing, China between June 16 - 20, 2013. Due to the rapid industrialization and urbanization during the past several decades, PM concentrations in Beijing are unusually high [14]. This led us to investigate the changes in cough sensitivity and pulmonary function among healthy volunteers from Sendai during their stay in Beijing. The principal aim of this study was to investigate the effect of short-term exposure to high PM concentrations on cough reflex thresholds, urge-to-cough, and pulmonary function in healthy subjects. The second aim was to assess the cough-specific health related quality of life before, during, and after a visit to a city with high levels of PM.
MATERIALS AND METHODS
Study Participants
We recruited 17 healthy volunteers (current smokers, n = 2; former smokers, n = 5; never-smokers, n = 10), with a mean age of 35 years (range 25-57 years) at the Tohoku University School of Medicine. The participants had no history of airway or pulmonary disease or respiratory tract infection for 6 weeks prior to the study period and during the study period. In order to avoid any effects on cough reflex thresholds, the participants were prohibited from smoking [15], eating, and drinking for at least 2 h prior to the test [16]. We obtained written informed consent from each participant, and the study was approved by the Ethics Committee of the Tohoku University School of Medicine (2013-1-9).
Study Design
The study comprised 3 test days. In order to prevent potential carryover effects from previous exposures, the tests were separated by 2 weeks [17, 18]. On the first day of testing, all measurements were conducted at Sendai, before the trip to Beijing, in order to establish a baseline. On the second day of testing, the participants were examined in the same order, over a 24 h period, after arriving at Beijing. The Security Export Control Office of the Tohoku University Hospital allowed us to export the device used in our research (4 June 2013). We did not restrict the movement of the participants, or the use of surgical masks was not restricted during their stay in Beijing. The average stay in Beijing was 4.0 days (range 2 - 8 days). All measurements were carried out under similar test conditions on the final day of testing, 2 weeks after returning to Sendai.
Air Quality Reports
Ambient PM2.5 and PM10 concentrations in Sendai were obtained from the Atmospheric Environmental Regional Observation System, which is controlled by the Ministry of Environment, Japan [19]. Ambient PM2.5 concentrations in Beijing were acquired from air quality monitoring data of the United States Embassy in Beijing [20]. Ambient PM10 concentrations in Beijing were obtained from Qingyue Open Environmental Data Center [21], which collates air quality data of the Chinese Ministry of Environmental Protection. The ambient PM2.5 and PM10 concentrations were expressed as the 24 h average on the day of testing at each of the study sites, according to the method described by Cakmak et al. [22]. As the environmental nitric oxide (NO) levels can affect fractional exhaled nitric oxide (FeNO) measurements [23], environmental NO was measured using an electrochemical sensor-based device (NIOX MINO, Aerocrine AB, Solna, Sweden) at the time of each test. Since the environmental NO levels in Sendai were below measurable limits (<5 ppb) on the days of testing, the average environmental NO level was obtained from the government database [19].
Cough Reflex Thresholds and Urge-to-Cough
The cough reflex challenge was performed using nebulized citric acid under normal tidal breathing with a nose clip. The nebulized solution was delivered through an ultrasonic nebulizer (NE-U17, Omron Co. Ltd., Kyoto, Japan) [24]. The details of the measurement process have been reported elsewhere [24, 25]. In brief, a series of citric acid solutions of varying concentrations, ranging from 0.7 g/L to 360 g/L, were delivered at two-fold incremental concentration intervals. In this study, the cough reflex threshold and suprathreshold were defined by the minimum citric acid concentration required to generate two or more coughs (C2) and five or more coughs (C5) in 1 min.
The participants graded their urge to cough using a modified Borg scale ranging from 0 (no need to cough) to 10 (maximum urge to cough). The citric acid concentration that resulted in the first perceivable urge to cough without associated motor cough was defined as Cu [26]. We plotted each subject's urge-to-cough scores against the corresponding citric acid concentrations using a log-log transformation. Linear regression analysis was used to calculate the slope on a log-log scale because of the linear relationship between the concentration of the tussive agent and the urge-to-cough scores [11, 27].
Pulmonary Function and Fraction of Exhaled Nitric Oxide
Pulmonary function evaluation was conducted to measure vital capacity (VC), forced expiratory volume in one second (FEV1), forced vital capacity (FVC), and peak expiratory flow (PEF) by using an electronic spirometer (Chestgraph Jr HI-101, CHEST, Tokyo, Japan), in accordance with the American Thoracic Society guidelines. We measured the concentrations of FeNO using an electrochemical sensor-based device. FeNO is considered to be an useful surrogate marker for eosinophilic airway inflammation and can be used to complement conventional physiological testing [28].
Cough-Specific Health Related Quality of Life
Cough-specific health related quality of life was assessed using the Leicester Cough Questionnaire-acute (LCQ-acute), which consists of 19 items divided into 3 domains: physical, psychological, and social [29]. The total score ranges from 3 - 21, with lower scores indicating greater impairment of health status due to cough. We obtained the necessary permission from the developer to use the instrument and to translate it from English into Japanese. The Japanese version of the LCQ-acute was developed by Dr. Satoru Ebihara (Toho University School of Medicine), Ryuhei Sato (Tohoku University Graduate School of Medicine; Kyoto University Graduate School of Medicine), Dr. Akio Niimi (Nagoya City University Graduate School of Medical Sciences), and Dr. Haruhiko Ogawa (Ishikawa-ken Saiseikai Kanazawa Hospital). The four Japanese researchers and the developer of the original version hold the copyright for the questionnaire.
Statistics
Data are expressed as mean ± standard deviation (SD), except when stated otherwise. Repeated measures were analyzed using repeated measures analysis of variance (ANOVA) followed by a Tukey’s multiple-comparison post hoc test. A two-tailed P-value of < 0.05 was considered significant. All analyses were performed using SPSS 17 software (SPSS Inc., Chicago, IL, USA).
RESULTS
The characteristics of the participants are summarized in Table 1.
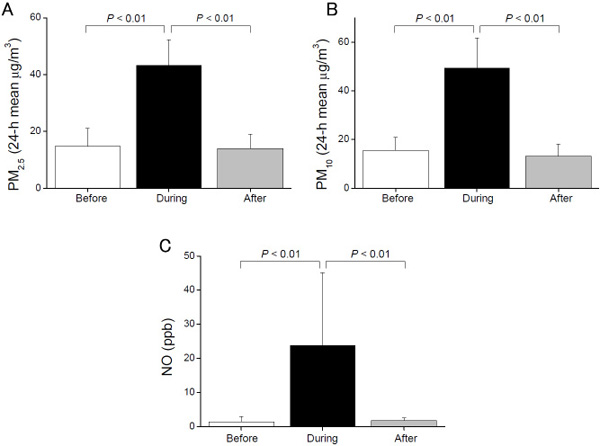
The PM2.5 concentration, on the day of testing, in Beijing (43.2 ± 9.1 μg/m3) was significantly higher than PM2.5 concentrations in Sendai, before (14.6 ± 6.4 μg/m3, P < 0.01) and after the trip (13.9 ± 5.1 μg/m3, P < 0.01) (Fig. 1A). Similarly, the PM10 concentration, on the day of testing, in Beijing (49.3 ± 12.4 μg/m3) was significantly higher than PM10 concentrations in Sendai, before (15.4 ± 5.4 μg/m3, P < 0.01) and after the trip (13.2 ± 4.8 μg/m3, P < 0.01) (Fig. 1B). The ambient NO level in Beijing was significantly higher than that in Sendai (P < 0.01 and P < 0.01, respectively) (Fig. 1C).
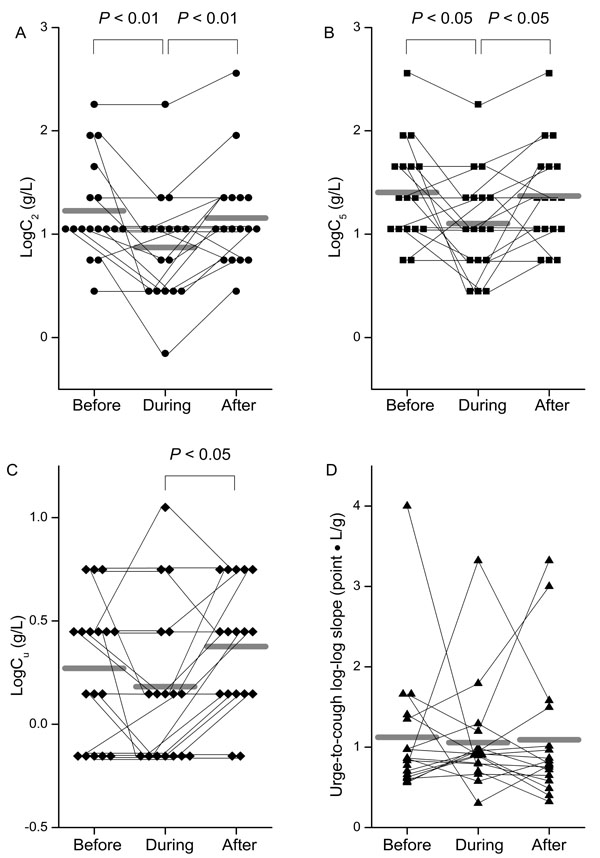
As shown in Fig. (2A), the cough reflex threshold to citric acid, expressed as log C2, during the stay in Beijing was significantly lower (0.87 ± 0.53 g/L) than in Sendai, before (1.23 ± 0.48 g/L, P < 0.01) and after (1.16 ± 0.50 g/L, P < 0.01) the trip. Similarly, log C5 during the stay in Beijing (1.10 ± 0.49 g/L) was significantly lower than before (1.40 ± 0.48 g/L, P < 0.05) and after (1.37 ± 0.49 g/L, P < 0.05) the trip (Fig. 2B). The urge-to-cough threshold, expressed as log Cu, was also significantly lower during the stay in Beijing (0.18 ± 0.38 g/L) than it was after the return to Sendai (0.38 ± 0.31 g/L, P < 0.05) (Fig. 2C), but there was no significant difference in the log-log slope of urge-to-cough between the 3 test days (Fig. 2D).
The results from the pulmonary function test and FeNO evaluation are shown in Table 2. Percentages of predicted VC, FEV1, and FVC, and FEV1/FVC were significantly lower in Beijing than on the pre-travel test day (P < 0.01, P < 0.01, P < 0.01, and P < 0.05). The FEV1 and FVC (percent predicted) improved significantly upon the return to Sendai in comparison to the measurements made in Beijing. There was no significant difference in the levels of FeNO at any time.
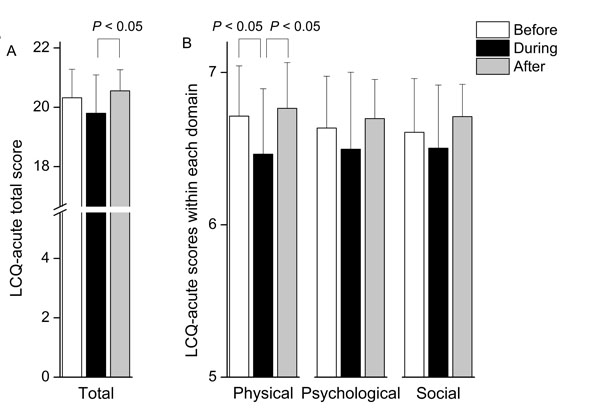
The total LCQ-acute score during the stay in Beijing (19.80 ± 1.29) was significantly lower than the score after the return to Sendai (20.55 ± 0.71, P < 0.05) (Fig. 3A). In particular, the physical health domain score during the stay in Beijing (6.46 ± 0.43) was lower than before (6.71 ± 0.33, P < 0.05) and after the trip (6.76 ± 0.30, P < 0.05) (Fig. 3B).
DISCUSSION
Our preliminary study presents three findings. Firstly, the cough reflex thresholds were significantly lower during the stay in Beijing, where PM concentrations were higher than they were in Sendai before and after the trip, and the urge-to-cough threshold during the stay in Beijing was significantly lower than it was after the trip. Secondly, impaired pulmonary function was observed during the stay in Beijing as indicated by reduced VC, FEV1, FVC, and FEV1/FVC. Finally, the reduced cough reflex and urge-to-cough thresholds and impaired pulmonary function during the short-term exposure to high concentrations of PM were accompanied by a reduction in subjective health-related quality of life due to cough scores.
The results of the present study concur with several previous studies that have reported an association between ambient PM concentrations and increased cough or impaired pulmonary function [6, 7, 14, 30, 31]. However, there is no previous study on the association between short-term exposure to particulate matter and cough reflex sensitivity or cognition of urge to cough. Studies performed under laboratory conditions have not reported the occurrence of cough or decreased pulmonary function associated with higher PM concentrations [12, 13]. The discordant results between the present study and the laboratory studies may be due to the extremely short exposure times, i.e., for 2 h, in the laboratory studies.
The cough reflex originates from the stimulation of sensory neuron receptors within the respiratory tract. TRPA1 and TRPV1, which are understood to contribute to the perception of the stimulus, have shown to be a part of the afferent sensory loop of the cough reflex [32]. Recent studies have shown that TRPA1 and TRPV1 are key mediators of the cellular response to environmental particulate matter [8-10]. Shapiro et al. [9] have shown that TRPA1 acts as a molecular sensor for wood smoke particulate matter in sensory neurons and human adenocarcinoma lung cells. In a study of the interaction between particulate matter and the cell surface, Deering-Rice et al. [10] reported that coal fly ash, which is an insoluble particulate matter, acted as a TRPV1 agonist. Based on these findings, it is possible to speculate that TRPA1 and TRPV1 activation in relation to the environmental particulate matter may be associated with the reduction in cough reflex and urge-to-cough thresholds that were observed during a short-term stay in Beijing, although the log-log slope of urge-to-cough did not differ between Beijing and Sendai. Since the urge to cough is a component of the brain's motivation system [33], this discrepancy suggests that the primary pathway for cough due to exposure to particulate matter may not involve higher centers, such as the cerebral cortex, but rather involves the brainstem cough control area [34].
The mechanisms underlying altered pulmonary function associated with particulate matter have been examined in several studies. Activation of TRPA1 by environmental particulate matter has been reported to be an important mechanism of pneumotoxicity [8, 9]. In addition, TRPV1 activation by particulate matter in airway and alveolar epithelial cells was shown to contribute to production of immunomodulatory cytokines and chemokines and promotes apoptosis in vitro [10, 35]. Thus, it appears that impaired pulmonary function caused by tissue damage and pulmonary inflammation is associated with both TRPA1 and TRPV1 activation, again potentially supporting a hypothesis that reduced cough reflex and urge-to-cough thresholds and impaired pulmonary function, observed in the present study, might have resulted from activation of TRPA1 and TRPV1 by environmental particulate matter. There was no significant difference in FeNO, which is a marker of airway eosinophilic inflammation, among the 3 test days. However, other studies have shown that humans exposed to concentrated ambient air particulate matter exhibit increased polymorphonuclear neutrophil counts [36]. Thus, our results indicate that airway inflammation by particulate matter arises from neutrophilic inflammation.
| Characteristic | |
|---|---|
| Number | 17 |
| Male/Female | 10/7 |
| Age (years) | 35.4 ± 9.6 |
| Height (cm) | 165.8 ± 7.3 |
| Weight (kg) | 58.4 ± 10.4 |
| Body mass index (kg/m2) | 21.1 ± 2.5 |
| Time from arrival in Beijing to second test (h) | 42.2 ± 13.5 |
| Variables | Before | During | After |
|---|---|---|---|
| Pulmonary function | |||
| VC (% predicted) | 110.7 ± 13.8 | 105.1 ± 12.6* | 108.2 ± 12.0 |
| FEV1 (% predicted) | 100.4 ± 13.2 | 91.7 ± 14.9*† | 96.8 ± 13.6 |
| FVC (% predicted) | 106.0 ± 10.9 | 100.4 ± 12.8*‡ | 105.5 ± 11.9 |
| FEV1/FVC (%) | 87.8 ± 5.8 | 84.6 ± 7.2§ | 85.1 ± 7.1 |
| PEF (% predicted) | 104.4 ± 11.8 | 97.7 ± 12.6 | 102.8 ± 15.6 |
| FeNO (ppb) | 19.9 ± 9.6 | 18.9 ± 10.1 | 22.0 ± 13.9 |
Regarding the reduced LCQ-acute scores during the stay in Beijing, an association between exposure to particulate matter air pollution and LCQ score has not been reported till date, previous studies have demonstrated that the LCQ score correlates with objectively counted coughs in subjects with chronic cough [37]. We surmise that the enhanced cough sensitivity among our subjects during their stay in Beijing, at a time of high PM levels, led to the impaired health status due to cough reflected by the LCQ-acute scores. However, we did not evaluate the characteristics of the cough (e.g. intensity, frequency) in the present study.
The strong point of this study is that acute effects of air pollution on respiratory health were investigated using various factors, including cough reflex and urge-to-cough thresholds, pulmonary function, FeNO, and LCQ-acute scores, allowing the problem to be viewed from different perspectives. However, our study has limitations. Ambient air pollution is a mixture of particulate matter and gases, which may also be associated with cough [38]. Moreover, other airborne particulates, such as pollen [39], fungus [40], or passive smoking [41], may affect results of this study because these are triggers for cough. Also, travelers’ fatigue could be a strong factor that affects the results of the pulmonary function test and cough reflex sensitivity test [42]. In addition, the ambient air pollution was not measured directly by us. Therefore, individual differences in exposure to particulate matter [43], and precise individual exposure estimates are unclear. Furthermore, the differences in the LCQ-acute total and LCQ-acute physical health domain scores, between the 3 days of testing, were low, despite being statistically significant [29], and the clinical importance of the differences remain unsolved, although we did use the Japanese version of the LCQ-acute, with permission from the developer. Finally, we recruited participants from the same city in Japan. In order to draw a more definitive conclusion, a larger sample study involving subjects from a wider area is warranted.
CONCLUSION
This pilot study provides data demonstrating that short-term exposure to PM concentrations, i.e., during travel, might induce changes in the cough reflex and urge-to-cough sensitivities, as well as in pulmonary function and health status due to cough. Our preliminary findings indicate acute changes in the respiratory system in a group of healthy volunteers during a stay in a city with high levels of ambient air pollutants. Exposure to particulate air pollutants in susceptible individuals, including children, the elderly, and patients with asthma or other chronic pulmonary diseases, may well pose an increased risk and this requires further investigation.
CONFLICT OF INTEREST
The authors confirm that this article content has no conflict of interest.
ACKNOWLEDGEMENTS
We thank Prof. Kefang Lai (State Key Laboratory of Respiratory Disease, Guangzhou Institute of Respiratory Disease, First Affiliated Hospital of Guangzhou Medical College) for valuable comments on the manuscript. This study was supported by Grants-in-Aid for Scientific Research from the Ministry of Education, Culture, Sports, Science and Technology, Japan (Grant numbers 25•7166, 23659375, 24300187, 24659397, 26460899, 15K12588); Research Grants for Longevity Sciences from the Ministry of Health, Labor and Welfare (H22-Junkanki-shi-Ippan-001) and Research Funding for Longevity Sciences (24-2) from National Center for Geriatrics and Gerontology (NCGG).


