All published articles of this journal are available on ScienceDirect.
Rare Unicentric Intrapulmonary Castleman Disease: A Systematic Review and Report of a Case
Abstract
Objectives
Castleman disease (CD) is a very rare B-cell lymphoproliferative disorder marked by the abnormal enlargement of lymph node tissue. It can present as either unicentric (UCD) or multicentric, with the former often appearing in intrathoracic regions, although its presence within the lungs is uncommon.
Methods
We report the case of a 42-year-old woman who underwent resection of an 11 cm intrapulmonary UCD. Additionally, we conducted a systematic review of the demographics, clinical presentation, diagnosis, and treatment approaches for intrapulmonary UCD.
Results
Our review identified 35 documented cases of intrapulmonary UCD, including our case. The average age was 34 years, with a female predominance of 57.7%. Tumor sizes ranged from 1.5 to 11 cm, with our case being the largest. Of the 24 cases with reported anamnesis, 58.3% were asymptomatic, while 41.7% had nonspecific symptoms such as cough, chest pain, or fever (as in our case). Histological analysis was available for 24 cases, with 83.3% identified as the hyaline vascular type. Biopsies through small needle aspiration or fresh-frozen samples failed in all attempts, requiring resection for diagnosis and treatment. Due to high vascularity, delicate location, and lack of diagnosis, lobectomy or pneumonectomy was performed in 45.7% of cases. Among the 11 cases with reported follow-up, no disease recurrence was observed over an average of 3 years.
Conclusion
Our systematic review highlights the rarity of UCD in the lungs. The demographics of intrapulmonary UCD align with the general disease profile. Surgical removal is crucial for both diagnosis and treatment. The significant vascularity and pulmonary location of these tumors present challenges, requiring pre-operative awareness and precautions.
1. INTRODUCTION
Castleman disease (CD) is a B-cell lymphoproliferative disorder marked by the abnormal growth of lymph nodes and, less commonly, extra-nodal locations (5%) [1]. Its rarity is underscored by an estimated 21 to 25 cases per million person-years, classifying it as an orphan disease [2].
CD presents in two distinct subtypes: the more common unicentric CD (UCD), usually asymptomatic, and the less common multicentric CD (MCD), which is often severe and systemic [3, 4]. Histologically, UCD can be divided into three main forms: a) hyaline vascular (HV) variant (76-91%), b) plasma cell (PC) variant (10-24%), and c) mixed variant (1-4%) [5].
The exact pathogenesis of CD remains unclear, but it is generally thought to result from impaired immunoregulation. Some theories suggest that UCD originates from an antigen stimulus affecting an abnormal plasmacytoid monocyte population in a lymph node, whereas MCD is believed to be triggered by factors such as chronic infection, chronic low-grade inflammation, viral infections, abnormal cytokine modulation, and, in some cases, infections with human herpesvirus-8 and HIV [3, 5].
The frequency of UCD is reported to be about 31% in the thorax (31, 29, 89%, respectively), 23% in the cervical region (6, 23, 42%, respectively), 18% in the abdomen (18, 38, 1%, respectively), with the remainder occurring more peripherally [1, 6, 7]. In intrathoracic cases, most affect either the mediastinum or the hilum [1, 6-10]. However, intrapulmonary locations, including those in the interlobar fissure, parahilar, and fully intraparenchymal areas, are considered exceptionally rare.
In this report, we provide a comparative analysis by systematically reviewing the limited previously published literature on intrapulmonary UCD. Additionally, we also present a rare case of intrapulmonary UCD.
2. CASE PRESENTATION
A 42-year-old Caucasian woman with a history of hay fever, asthma, metabolic syndrome, and prior radiotherapy for thyroid adenoma consulted her general practitioner due to a persistent cough and fever lasting three weeks. During the consultation, her thyroid and inflammatory markers appeared normal, but lung auscultation revealed rales in her left hemithorax. A chest x-ray showed an indistinct contour in the middle left lung area.
A subsequent thorax CT scan (Fig. 1A-D, F) revealed an 11 x 8 x 5 cm partially necrotic mass with cystic, adipose, slightly calcified characteristics, and contrast agent uptake. The mass extended from the mediastinum along the left hilus, the junction of the left main bronchus, and into the left lung. There was an extensive connection between the pars interlobaris of the pulmonary artery and the upper and lower pulmonary veins on the left side. Although there was no apparent direct invasion of these blood vessels, potential diagnoses included primary lung cancer, angiosarcoma, ectopic thyroid tissue, or a teratoma (though the location was atypical).
A PET-CT scan with FDG enrichment showed minimal metabolic activity within the mass (Fig. 1E), making lung cancer, angiosarcoma, and thyroid tissue involvement less likely. This shifted the differential diagnoses toward a neuroendocrine tumor or a solitary fibrous tumor of the pleura. A transbronchial needle biopsy was non-conclusive, revealing fibrotic, non-malignant cells.
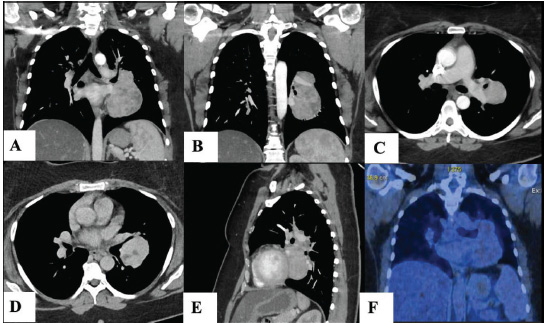
CT scan of the Unicentric Castleman tumor A) hemi-encircling the left upper lobe bronchus in coronal view, B) the inflow into the upper- and lower pulmonary vein in coronal view, C) the attachment and nutritive branch from the pars interlobaris of the pulmonary artery in axial view, D) the broad attachment in the bifurcation of the upper and lower lobe bronchus in axial view, E) the location with neighboring vessels and bronchi in sagittal view. F) PET-CT scan showing slight metabolic activity in the tumor.
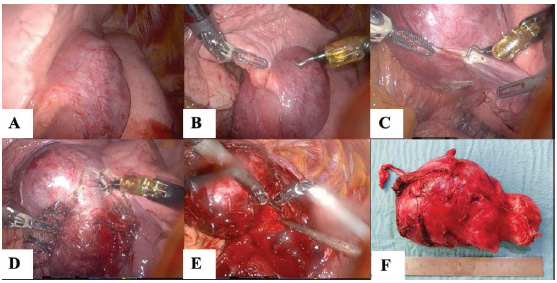
Unicentric castleman tumor during robotic assisted thoracic surgery A) view on interlobar fissure, B) resection from upper lobe, C) resection from lower lobe, D) resecting branches draining into the lower lobe veins, E) view on the pars interlobaris of the pulmonary artery in the opened interlobar fissure. F) Resected histological specimen.
Following an interdisciplinary board discussion, the patient underwent a diagnostic DaVinci robot-assisted thoracoscopy (Fig. 2A-E). During the procedure, it was discovered that the tumor was unattached to the mediastinum and hilum but located in the pulmonary fissure, attached by lung tissue from the upper and lower lobes, partially encircling the upper lobe bronchus. It was supplied by a branch from the pulmonary artery and bronchial artery and drained into the upper and lower lobe veins. A frozen section biopsy failed to provide a conclusive diagnosis, as it contained some lymphatic cells, which could potentially indicate a lymphatic neoplasia. Consequently, the surgical team proceeded with the resection and mobilization of the mass while preserving its lung tissue capsule.
Due to significant vascularity toward the center of the fissure and insufficient exposure for precise vessel ligation, the surgical approach was converted to a thoracotomy. The tumor was completely removed while sparing all lung tissue. However, due to ongoing bleeding, with a total blood loss of 2000 ml, a large hemostasis patch (VerisetTM 8x16cm) was applied to the wound bed, resulting in rapid and successful hemostasis. The entire surgical procedure concluded after 145 minutes.
Following the surgery, the patient received a transfusion of 2 units of red blood cells. She was extubated just one hour after the procedure and transferred from the intensive care unit to the regular ward the following day. Her subsequent hospital stay was without significant complications, and she was discharged on the sixth day post-operation.
The removed surgical specimen weighed 133 grams (Fig. 2F). The conclusive histological examination (Fig. 3A-C) revealed a large lymph node with lymphoid tissue showing angio-follicular hyaline hyperplasia, numerous regressed germinal centers, interstitial vascular proliferation, and increased interstitial fibrosis. Immunohistochemical studies showed no signs of lymphoma. After consultation with a second pathologist, the diagnosis of CD, specifically the localized hyalin vascular variant, was confirmed. There was no evidence of malignancy, and the resection was considered radical and in healthy tissue.
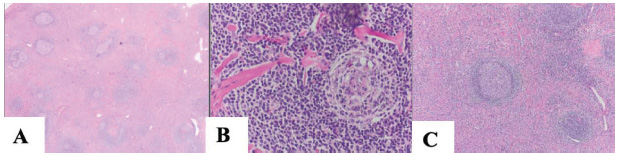
Histological slides of the resected unicentric Castleman tumor of the hyaline vascular type: A) overview (HE x40), B) a typical hyalinized vessel radially penetrating the germinal center (lollipop sign) (HE x100), C) lymphoid follicles with atrophic germinal centers showing proliferation of follicular dendritic cells (HE x100).
Since there was no radiological evidence of additional lesions, it was determined that this form of CD was unicentric. The complete removal of this single lesion was deemed curative, so the interdisciplinary medical board decided against further treatments, opting for regular follow-ups instead.
During the first follow-up after six months, a CT scan showed no signs of relapse, and the patient remained symptom-free. It was then decided to extend the interval, until the next follow-up, to one year.
3. METHODS
In April 2024, a systematic review was conducted in accordance with the PRISMA guidelines. The PubMed database was used to identify case reports and case series that evaluate the demographics, clinical presentation, diagnosis, and treatment strategies of intra-pulmonary UCD. Additionally, a non-systematic search of the Google Scholar database was conducted using the same keywords.
The following search string was used:
((“Castleman”[tiab] OR “Morbus Castleman”[tiab] OR “Castleman’s disease”[tiab] OR “Castleman disease “[tiab] OR “lymphoid hamartoma”[tiab] OR “lymph node hyperplasia”[tiab]) AND (“lung”[tiab] OR ” pulmonal”[tiab] OR “intrapulmonal”[tiab] OR “intra-pulmonal”[tiab] OR “pulmonary”[tiab] OR “intra-pulmonary”[tiab] OR “intra-pulmonary”[tiab] OR “bronchial”[tiab]))
Promising original articles and reviews were also scrutinized for any additional relevant articles listed in their references. The inclusion criteria focused on intrapulmonary CD without restrictions regarding patient age, publication year, or language; all foreign language articles were translated and assessed via Deepl.com. Cases of MCD with pulmonary lesions and cases of UCD reported at the pulmonary hilus or mediastinum were excluded.
The database search and individual article reviews were independently conducted by both M.K.P. and J.E.P. to prevent selection bias. The studies identified were independently organized according to the specific sub-criteria outlined in Table 1. These tabulated studies were then compared to identify any potential disagreements.
The results are presented as means and percentages, either for the total number of identified cases or for specific subgroups or sub-criteria. Studies that lacked data on a particular sub-criterion were excluded from the analysis of that sub-criterion.
| Study/Refs. | Location | Size (cm) | Histology | Diagnostics | Treatment | Age | Sex | Symptoms | Onset | Disease-Free Time |
|---|---|---|---|---|---|---|---|---|---|---|
| Current Case | Parahilar LUL & interlobar fissure | 11x8x5 | HV | Transbronchial biopsy; intra-OP fresh-frozen section (non-diagnostic) | Lung resection (RATS switched to open) | 42 | F | Cough, fever | 3w | ≥ 6m |
| Keller 1972 [6] A | Parahilar, adjacent to a bronchus | NS | Likely HV | NS | Open lobectomy/pneumonectomy | NS | NS | NS | NS | NS |
| Keller 1972 [6] B | Parahilar, adjacent to a bronchus | NS | Likely HV | NS | Open lobectomy/pneumonectomy | NS | NS | NS | NS | NS |
| Keller 1972 [6] C | Intraparenchymal (coin lesion) | 1.5 | Likely HV | NS | Open lobectomy/pneumonectomy | NS | NS | NS | NS | NS |
| Awotedu 1990 [34] | Intraparenchymal LLL, segment 6 | 8x6 | HV | Pleural biopsy (inflammatory changes) | Open pneumonectomy | 21 | M | Chest pain, fever, effusion | 2w | ≥ 3y |
| Lee 1991 [35] | Parahilar ML | NS | HV | NS | NS | NS | NS | NS | NS | NS |
| Caus 1992 [36] | Interlobar fissure | NS | NS | NS | NS | NS | NS | NS | NS | NS |
| Spendini 1995 [37] | Interlobar fissure ML & RLL | NS | NS | Transbronchial biopsy (non-diagnostic) | Surgical resection | 51 | F | NS | NS | ≥ 3y |
| Leung 1997 [28] | Interlobar fissure ML & RLL | NS | HV | NS | VATS excision | 30 | F | Asymptomatic, anemia | - | ≥ 19m |
| McAdams 1998 [8] | Parahilar LLL, interlobar fissure | ~10 | HV | NS | Open resection | 28 | M | Asymptomatic | - | NS |
| Ferrozzi 2001 [15] | Intraparenchymal LUL, segment 3 | 1.5 | HV | NS | Tumor excision | 46 | F | Asymptomatic | - | NS |
| Hosoda 2003 [12] | Left interlobar fissure | NS | NS | NS | Open resection | 21 | M | Asymptomatic | - | NS |
| Nishii 2004 [27] | Right interlobar fissure | 3x2x2 | HV | NS | VATS removal | 33 | M | Asymptomatic | - | NS |
| Minami 2005 [38] | Left interlobar fissure | 7x5.5x4.5 | NS | NS | Open extirpation | 19 | F | Cough | NS | NS |
| Mohanna 2006 [11] | Intraparenchymal LUL, lingula | 4x3.5 | HV | No | Complete surgical resection | 54 | F | Back pain | 1m | ≥ 14y |
| Yeh 2007 [25] | Parahilar LUL | 4.5x3 | HV | No | LUL resection | 42 | M | Cough | 2m | NS |
| Kawczun 2007 [39] | Interlobar fissure RUL & ML | 3 | HV | Pre-op angiography, intra-OP fresh-frozen section (malignancy suspected) | Open resection | 40 | F | Chest pain, dyspnea | 20y | NS |
| Yekeler 2009 [21] | Interlobar fissure ML & RLL | 3.5 | HV | No | Open resection | 43 | F | Cough | 6m | NS |
| Racil 2009 [18] | Interlobar fissure RUL & ML | 4.4x2.8 | HV | No | Pneumonectomy | 23 | F | Dyspnea, cough | 4y | ≥ 1y |
| Tokunaga 2009 [14] | Parahilar LLL | 3.5 | HV | No | LLL resection | 23 | F | Asymptomatic | known≥ 7y | NS |
| Wang 2009 [40] | Parahilar LLL, interlobar fissure | 7.5 | HV | No | Tumor excision | 27 | M | Asymptomatic | - | NS |
| Günlüoglu 2011 [41] | Parahilar RLL & interlobar fissure | 5.5 | PC | Transbronchial, transthoracal biopsy (inconclusive) | Open limited resection | 29 | M | Asymptomatic | - | NS |
| Murinello 2011 [42] | Intraparenchymal RLL, segment 6 | 2 | PC | No | RLL resection | 60 | M | Asymptomatic | - | ≥ 9m |
| Ota 2013 [22] | Parahilar RLL | 5 | HV | No | VATS resection RLL | 19 | M | Asymptomatic | - | ≥ 8m |
| Chiang 2013 [43] | Intraparenchymal LUL, lingula | 2.1x1.2 | PC | NS | Wedge resection | 72 | F | Asymptomatic | - | NS |
| Nadir 2014 [17] | Parahilar RUL | 6x5x4 | HV | No | Open resection RUL | 28 | F | Asymptomatic | NS | NS |
| Rawashdeh 2015 [13] | Intraparenchymal LUL | 4.8 | HV | Intra-OP frozen section (able to rule out carcinoma) | VATS LUL resection | 16 | F | Chest pain, cough | 3y | NS |
| Liu et al 2014 [44] | LUL (likely intraparenchymal) | 3.1x2.9x3 | HV | No | LUL resection | 32 | M | Asymptomatic | - | ≥ 2y |
| Luo 2015 [9] | intrapulmonary fissure | NS | Likely HV | NS | Open resection | NS | NS | NS | NS | NS |
| Luo 2015 [9] | lung parenchyma | NS | Likely HV | NS | Open resection | NS | NS | NS | NS | NS |
| Haager 2015 [26] | Parahilar ML & interlobar fissure | 3x2.4 | HV | Transbronchial biopsy (non-diagnostic). Intra-OP frozen section (lymphoma/carcinoid appearing) | Open resection ML | 24 | M | Cough, bronchopulmonary infection | <1m | NS |
| Cao 2015 [45] | RLL | 4.5x3.5x3 | MC | Bronchoscopic biopsy (lymphocytic tissue) | Tumor excision | 39 | F | NS | 2m | ≥ 6m |
| Sarana 2017 [24] | Interlobar fissure RUL & ML | 2.5x2.5x2 | HV | No | VATS switched to lung-sparring thoracotomy | 22 | F | Asymptomatic | known≥ 2y | ≥ 6y |
| Xiaoxian 2020 [10] | parahilar partially in lung parenchyma | NS | Likely HV | NS | VATS lobectomy | NS | NS | Asymptomatic | NS | NS |
| Xiaoxian 2020 [10] | Intraparenchymal | NS | Likely HV | NS | VATS lobectomy | NS | NS | Asymptomatic | NS | NS |
4. RESULTS
The systematic review conducted in PubMed yielded 318 publications from 1970 to 2024. After screening titles and abstracts, 81 articles were selected for further examination, including their reference lists. Forty-four studies with a total of 132 reported cases of MCD with pulmonary lesions were excluded, along with 11 records that did not meet the pulmonary UCD criteria. Two additional records were identified through reference lists. No conflicts or disagreements arose between the two authors who independently reviewed the database.
Additionally, a non-systematic search of the Google Scholar database revealed two more records. This process resulted in a total of 30 articles reporting 34 cases. The literature selection process is displayed in the PRISMA flow diagram (Fig. 4). Table 1 summarizes the collected cases.
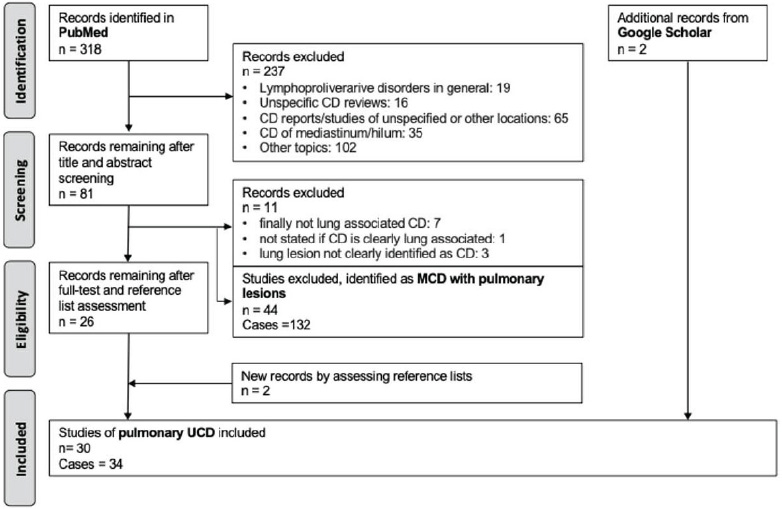
PRISMA diagram investigating resources on unicentric Castleman disease. A total of 318 articles were retrieved from PubMed. During the initial screening process, 237 articles which did not meet the inclusion criteria were excluded, leaving 81 articles included. A second screening process resulted in 11 and 44 articles being excluded, leaving 26 full length articles. During the final phase of evaluation, a total of four extra articles were discovered from reference lists and Google Scholar, resulting in 30 pulmonary UCD articles and 34 cases.
The reporting among the 34 cases was highly heterogeneous. Complete datasets that met all the desired sub-criteria listed in Table 1 were available for only 7 of the 34 cases. The analyses of these sub-criteria are presented in the discussion section.
5. DISCUSSION
Intrapulmonary UCD is rare. Our presented case, along with the systematic review, revealed a total of only 35 documented intrapulmonary UCD cases (Table 1).
5.1. Prevalence/Incidence
Estimating the frequency of intrapulmonary UCD within the broader context of all UCD localities is challenging due to its status as an orphan disease. However, intrapulmonary UCD appears to occur in a low single-digit percentage range. For instance, Keller et al. (1972) [6] reported an intrathoracic occurrence of 89.2% (66/74) in a large series of 74 UCD patients, but only 2.7% (2/74) had isolated lung involvement. In contrast, a systematic review by Talat et al. (2012) [7] found a 28.1% intrathoracic occurrence in 235 cases, with no instances of lung involvement. Similarly, Danon et al. (1993) [1] reported a 31% intrathoracic occurrence among 102 patients but no lung involvement. Additionally, several other studies reflect the rarity of intrapulmonary UCD. McAdam et al. (1998) [8] reported a 4.2% (1/24) intrapulmonary occurrence in their series on intrathoracic UCD. Lou et al. (2015) [9] found a 12.5% (2/16) occurrence, and Xioxian et al. (2020) [10] reported an 11.1% (2/22) occurrence in their respective case series. These findings collectively indicate that intrapulmonary UCD is indeed quite uncommon.
5.2. Histology
Among the 35 identified intrapulmonary UCD cases, 24 had clearly reported histological subtypes and seven more were likely of the HV subtype. Of the cases with clear histology, 83.3% (20/24), were of the HV type, including our case, 12.5% (3/24) were of the PC type, and 4.2% (1/24) were of the MC type. The predominance of the HV type was also observed in the case series by McAdam et al. (1998) [8] and Lou et al. (2015) [9], which reported 95.8% and 75% HV type for various intrathoracic localities, respectively. This aligns with the typical histology of UCD in general, estimated to be 77.7% in the systematic review by Talat et al. (2012) [7] and even 91% in the large case series by Keller et al. (1972) [6].
5.3. Symptoms
In general, UCD is often reported to be asymptomatic (51-66.6%) [4, 6] or to present with flu-like symptoms [5, 11]. Among the 35 compiled cases of intrapulmonary UCD (Table 1), only 24 cases provided information on symptom anamnesis. Of these, 14 (58.3%) were entirely asymptomatic, while 10 (41.7%) exhibited nonspecific symptoms such as cough (N=7/10), fever (N=2/10), dyspnea (N=2/10), and chest or back pain (N=4/10). The coughing symptoms may be partly explained by compression and irritation of the adjacent bronchial system or post-stenotic infection, and the pain may be due to pressure on the pleura parietalis. In three cases, the intrapulmonary lesion was observed for 1 [12], 3 [13], and even 7 years [14] before resection, showing varying growth dynamics from very slow to tripling in size within a year. The HV subtype's ability to grow slowly over many years has been described in other localities [1].
5.4. Location
No side specificity for intrapulmonary UCD was detected in our literature review. Of the documented cases, 13 UCD lesions were located in the right lung and 14 in the left lung, while the rest did not specify. Regarding the location, 9 cases were described as fully embedded in the periphery of the lung parenchyma, 13 cases had lesions originating parahilar, proximal on a lobe bronchus, sometimes expanding into the interlobar fissure as in our case, 11 cases originated in the interlobar fissure itself, and 2 cases did not provide details.
5.5. Lesion Size
Among the cases listed in Table 1, the average size of the pulmonary lesions was 4.62 cm, with sizes ranging from 1.5 cm to 11 cm. Our reported case is noteworthy as it stands out as the largest intra-pulmonary CD reported to date. Such a large size of UCD appears uncommon compared to the documented growth potential in other body locations. In large series, Danon et al. (1993) [1] and Keller et al. (1972) [6] reported medians of 7 cm and 5.8 cm and maximum sizes of 10 cm and 16 cm, respectively. Talat et al. (2012) [7] detected a mean size of 5.5 cm in their large review of intrathoracic cases, with a maximum of up to 20 cm, while McAdam et al. (1998) [8] even reported an outstanding intrathoracic lesion size of 25 cm.
5.6. Age
Among our compiled intrapulmonary UCD cases, the average age at presentation was 34.0 years, ranging from 16 to 72 years. Generally, UCD can occur at any age [5], but our finding aligns closely with the mean age of 33.8 ± 17.8 years found in a systematic review of 278 cases [7] from various locations, the mean age of 30.5 ± 8 years in a case series of 32 intrathoracic UCD cases [9], and the mean age of 34 years in a case series of 24 intrathoracic UCD cases [8].
5.7. Gender
Regarding gender in our detected intrapulmonary UCD cases, 15 were identified as female and 11 as male, while gender was not stated in 9 cases. Excluding the unstated group, the proportion was 57.7% female to 42.3% male. Female dominance was also reported in various UCD locations in the review by Talat et al. (2011) [7], which found 59.4% (165/278) female, and the case series of Danon et al. (1993) [1], which reported 66% (71/93) female. Similarly, the intrathoracic series by McAdams et al. (1998) [8] reported 75% (18/24) female [8]. However, the case series by Keller et al. (1972) [6] for various locations and the case series by Luo et al. 2015 [9] on various intrathoracic locations found no particular gender dominance, with the latter reporting an equal distribution of 50% (8/16) female.
5.8. Ethnicity
No race predominance was observed in our intrapulmonary UCD series. Of the collected cases, 42.8% (15/35) were of Asian origin, which aligns with the global population distribution. Similarly, Keller et al. (1972) [6] did not observe any obvious race predominance in their series involving various localities
5.9. Imaging
Among all intrapulmonary UCD reports, no typical pathognomonic characteristics were detected in CT, PET-CT, or MRI scans. This is a known issue for CD in general, leading to various differential diagnoses [7, 8, 15-17]. Additionally, only subtle correlations exist between radiological findings and histological types [15, 16, 18].
5.10. Diagnosis
Histopathological assessment of a large tumor biopsy is the gold standard for CD diagnosis [19, 20]. All six reported cases where an intrapulmonary UCD lesion was attempted to be diagnosed solely by bronchoscopic small needle aspiration or transthoracic biopsy were inconclusive. Notably, in five cases, including ours, intraoperative fresh-frozen sample diagnosis was also inconclusive. Furthermore, biopsies may pose a high bleeding risk due to the typically highly vascularized nature of CD tumors [21, 22], a scenario specifically reported in one case [18].
5.11. Management
Complete surgical resection is generally considered the gold standard for managing UCD [19, 23]. However, our review revealed that resecting intrapulmonary lesions is particularly delicate and demanding.
Out of the 35 compiled intrapulmonary UCD cases listed in Table 1, truly lung-sparing, limited resections that preserved most of the lung parenchyma were clearly reported in only two cases (5.7%): the case of Sarana et al. (2017) [24] and our case.
At least 45.7% (16/35) of the intrapulmonary UCD cases required significant lung parenchyma resection. Specifically, 11 cases (31.4%) involved lobectomy, 3 cases involved either lobectomy or pneumonectomy and 2 cases definitely required pneumonectomy. However, there were likely more instances of significant resection that were not detected due to the lack of reporting.
The reasons for extensive lung resection were stated as follows: A) the necessity to be radical due to the inability to intraoperatively exclude pulmonary malignancy [14, 17, 25], B) the central intraparenchymal location of the lesion [26], and C) the rich blood supply of the HV type leading to severe intraoperative bleeding [18, 22, 24]. Excessive hemorrhage is a well-known issue reported from several lesions in other intrathoracic locations: Keller et al. (1972) [6] and McAdam et al. (1998) [8] reported 4 and 6 such cases, respectively. Both we and Sarana et al. (2017) [24] also encountered high vascularity, which necessitated abandoning the minimally invasive robotic and thoracoscopic methods, respectively, in favor of thoracotomy.
In only 6 of the compiled cases (17.1%) [10, 13, 22, 27, 28], the intrapulmonary UCD lesions, up to 5cm in size, were specifically described as having been removed via minimally invasive thoracoscopy. The other 82.9% of cases required thoracotomy for sufficient visibility. The challenge of successful minimally invasive resection for UCD lesions is not limited to the lung. Talat et al. (2012) [7] reviewed various intrathoracic UCD cases and found that only 3 out of 93 cases (3.2%) were operated on thoracoscopically. Similarly, Lou et al. (2015) [9] reported that only 1 out of 16 cases (6.3%) underwent thoracoscopic surgery.
Regarding the radicality of resection of intrapulmonary UCD, Sarana et al. (2017) [24] reported a subtotal resection due to bleeding and lung-sparing intentions. Most other intrapulmonary UCD reports did not specify their microscopic resection margins, but the number of subtotal resections might be high. For instance, McAdam et al. (1998) [8] reported subtotal resections in 10 out of 24 cases of intrathoracic, non-lung UCD localities due to the challenge posed by adjacent vessels or other delicate intrathoracic structures. As a consequence, surgeons should be mindful of the heightened risk of intraoperative severe bleeding, which is especially notorious in the hyaline vascular type. To mitigate such complications, preoperative angiography of the lesion to understand its blood supply is recommended [22], and angioembolization has occasionally been reported for intrathoracic, though not specifically for intrapulmonary lesions [29-31].
Rather than surgery, a few authors recommend radiotherapy as an alternative, particularly in cases where surgical intervention is impractical or if the lymph node is inaccessible [20, 23]. However, we did not find any instances of intrapulmonary UCD in our literature review that were treated exclusively with radiotherapy. Only one case [24], which was believed to have been removed non-radically, received adjuvant stereotactic radiation and showed no recurrence within at least six years. In a study involving 32 patients with UCD or MCD in various locations, treating solely with radiotherapy resulted in a complete response rate of only 43.8%, a partial response rate of 43.8%, and a non-response rate of 12.4%, with no observed dose-response relationship. The authors concluded that neoadjuvant radiotherapy might facilitate surgical resection of locally advanced or irresectable UCD disease. However, they did not recommend radiotherapy as an alternative to surgery [32].
5.12. Prognosis
The disease-free survival of intrapulmonary CD after resection was scarcely reported in our literature analysis. Only 11 out of 24 intrapulmonary UCD cases included follow-up data, ranging from 6 months to 14 years (mean 3.0 years), with no reported recurrences. A systematic review of 278 patients with UCD at various locations found that surgical tumor resection resulted in an impressive 95% disease-free survival rate at 3 years [7].
5.13. Intrapulmonary Multicentric CD
Of note, despite the rarity of UCD in the lung, our literature review frequently reported lung involvement in MCD. We identified 132 cases of MCD with pulmonary lesions, indicating that 21.0% of pulmonary Castleman cases are UCD, while 79.0% are MCD with intrapulmonary involvement. This suggests that pulmonary Castleman disease is more likely to be of the MCD type than the UCD type. A large case series on 162 patients with MCD found a pulmonary involvement rate as high as 35.8% [33]. However, considering the overall estimated occurrence of MCD at only about 23% [2], this is remarkable. It shows an opposite trend in the lung compared to other parts of the body, where UCD is more common (Table 1).
CONCLUSION
Our systematic literature review highlights the extreme rarity of intrapulmonary UCD, with only 35 reported cases. In contrast, MCD with lung involvement is more frequent. Due to the limited number of cases and the often-incomplete reporting, comparisons and definitive conclusions are challenging. However, the demographic characteristics for intrapulmonary UCD appear to align broadly with the general disease profile. Resection is essential for diagnosis and is the gold standard for successful long-term therapy. The lung location is particularly challenging, and the high vascularity of these tumors must be considered to avoid significant blood loss and to achieve lung-sparing surgery.
AUTHORS' CONTRIBUTION
It is hereby acknowledged that all authors have accepted responsibility for the manuscript's content and consented to its submission. They have meticulously reviewed all results and unanimously approved the final version of the manuscript.
LIST OF ABBREVIATIONS
| CD | = Castleman Disease |
| PC | = Plasma Cell |
| MCD | = Multicentric CD |
AVAILABILITY OF DATA AND MATERIALS
The data and supportive information are available within the article.
ACKNOWLEDGEMENTS
Declared none.


