All published articles of this journal are available on ScienceDirect.
Monitoring Patient/Ventilator Interactions: Manufacturer’s Perspective
Abstract
The introduction of reduced and more powerful electronics has allowed the transition of medical equipment such as respiratory support devices from the hospital to the patient’s home environment. Even if this move could be beneficial for the patient, the clinician ends up in a delicate situation where little or no direct supervision is possible on the delivered treatment.
Progress in technologies led to an improved handling of patient-device interaction: manufacturers are promoting new or improved ventilation modes or cycling techniques for better patient-ventilator coupling. Even though these ventilation modes have become more responsive to patient efforts, adversely they might lead to events such as false triggering, autotriggering, delayed triggering.
In addition, manufacturers are developing tools to enhance the follow-up, remotely or offline, of the treatment by using embedded memory in the respiratory devices. This logging might be beneficial for the caregiver to review and document the treatment and tune the settings to the patient’s need and comfort. Also, remote telemedicine has been raised as a potential solution for many years without yet overall acceptance due to legal, technical and ethical problems.
Benefits of new technologies in respiratory support devices give the technical foundation for the transition from hospital to home and reducing patient/ventilator asynchronies. Healthcare infrastructure has to follow this trend in terms of cost savings versus hospital stays.
MECHANICAL VENTILATION: FROM HOSPITAL TO HOME
The main objectives for mechanically ventilating a patient are to improve oxygenation, to increase or maintain minute ventilation, to help CO2 clearance, yet by decreasing work of breathing and protecting the airways.
Mechanical ventilators should be adjusted to influence the breathing rate and tidal volume by taking into account patient’s physiology and interfaces’ dead space.
In the Intensive Care Unit, common Indications for ventilation were reviewed by an international group in 2000 [1],
- Hypoxemic respiratory failure 66%
- Acute exacerbation of COPD 13%
- Neuromuscular disorders 10%
- Coma 10%
There is a trend changing towards Non-Invasive Ventilation (NIV) [2-4], especially in the intermediate care units [5] and even more in the home environment [6]. This trend is described in the European survey of 2002: with improving patient status from the Intensive Care Unit to the Respiratory Intermediate Care Unit and the finally the Respiratory Monitoring Unit, more NIV is used [5].
Even if this epidemiology study mainly considers intermediate care units and not home care, home-care environment as place of care has quickly moved towards Non-Invasive Ventilation use [7-9].
The epidemiology of home mechanical ventilation was described in the EuroVent study [8]. This study reviewed the usage of home mechanical ventilation in 329 centres in 16 European countries including more than 16509 patients. The authors concluded wide variations in experience and care provision infrastructures between countries (Fig. 1).
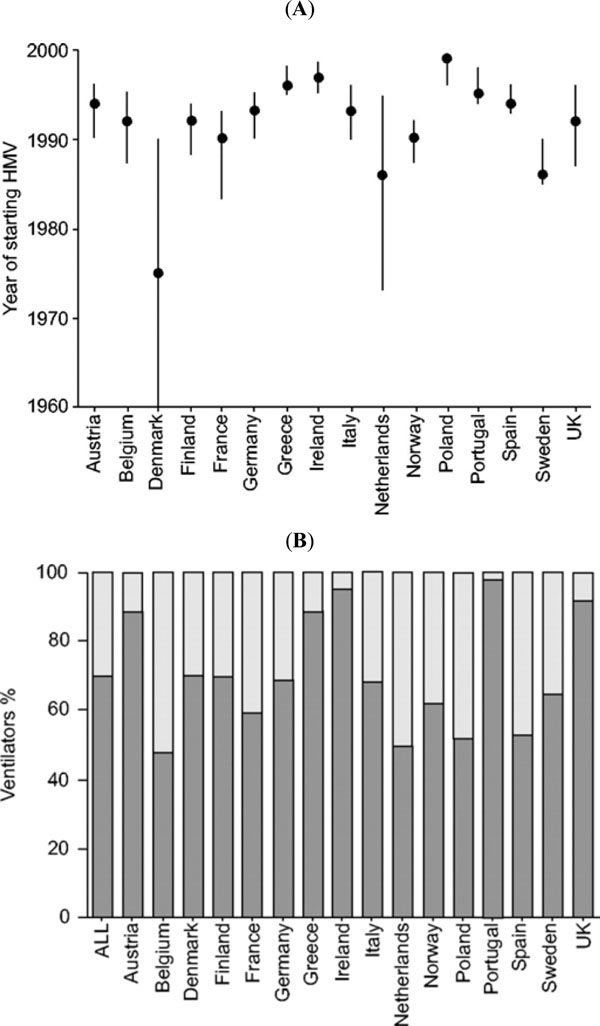
(A) Median year of starting home mechanical ventilation; (B) Percentage of ventilator type (black: pressure; grey: volume) [8].
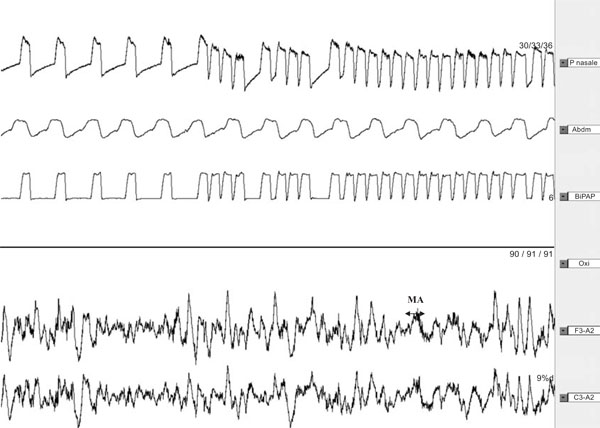
Polysomnography recording (2min) of a subject with normal ventilation under NPPV during slow-wave sleep [14].

Polysomnography recording (2 min) of asynchrony in slow wave sleep with micro-arousals [14].
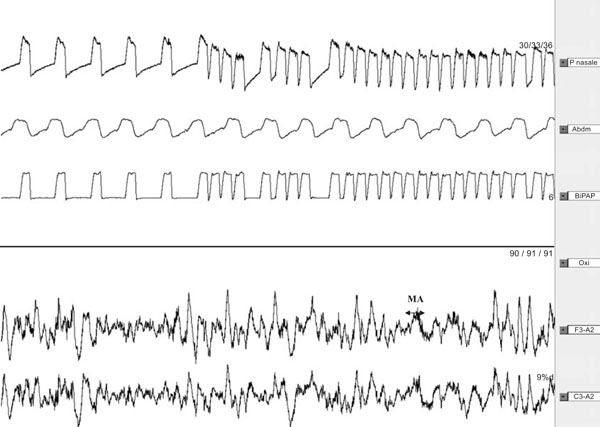
Polysomnography recording (2min) with auto-triggering during slow wave sleep, inducing microarousals [14].
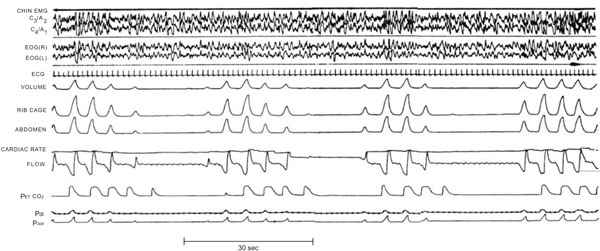
Polysomnography recording showing Periodic Breathing with repetitive central apnoeas [38].
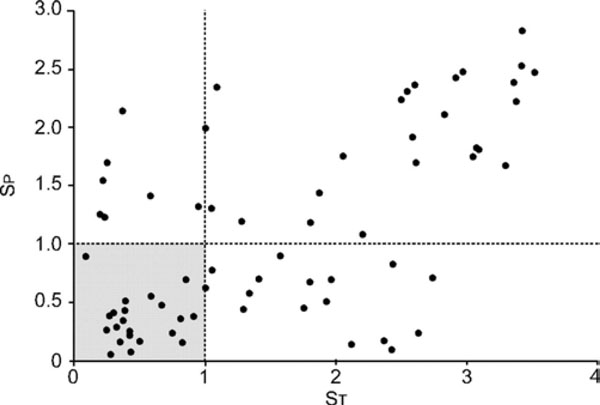
Graph showing patient–ventilator interactions during NIV. ST: Shannon entropy related to ventilatory variability; SP: Shannon entropy related to ineffective triggering efforts (optimal zone SP and ST<1).
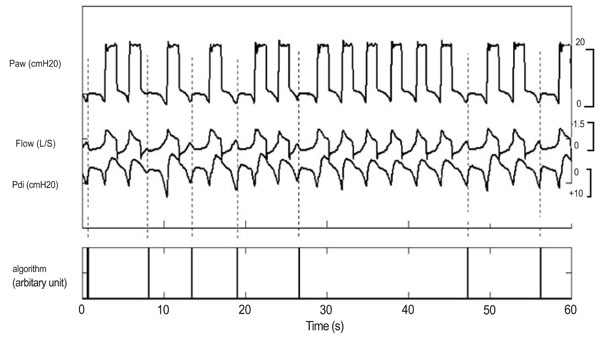
Traces showing ineffective breaths during PSV [16].

Various commercially available devices for establishing remote connections.

Troubleshooting of crucial issues in pressure support ventilation [67].
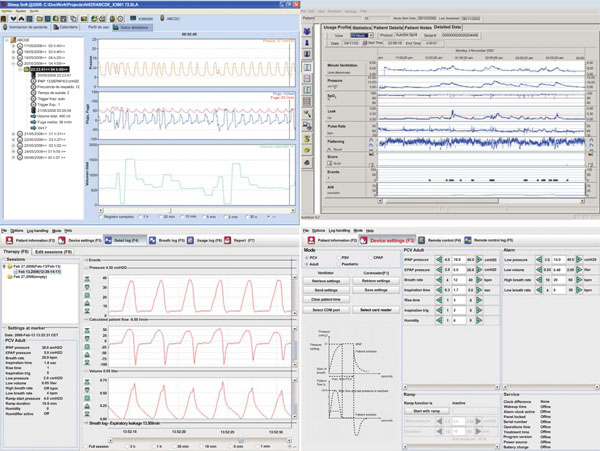
Screenshots of 3 commercially available software packages to log patient/ventilator interaction or change settings online or remotely.
After weaning from invasive ventilatory support and if the patient still requires additional support, long-term home non-invasive ventilation is a key choice for treating hypercapnic chronic respiratory failure patients in absence of socio-economic or medical constraints [10]. Clini and colleagues reviewed data from Italy comparing home NIV (with oxygen) with the cost care of oxygen alone. Hospital related costs were lower in the NIV group [11-13].
Using NIV in the home puts more stress on finding the appropriate settings for the patient on a specific ventilator. Successful ventilation does depend on reduces patient ventilator asynchronies. Asynchronies or more generally patient/ventilator respiratory disturbances may occur and may contribute to failure or discomfort.
PATIENT VENTILATOR INTERACTIONS
Guo and colleagues identified the patient/ventilator respiratory disturbances to be relatively frequent [14]. They described 4 types of respiratory events versus normal synchronous respiration on a ventilator (Fig. 2):
- patient/ventilator desynchronization (Fig. 3),
- periodic breathing (Fig. 5),
- autotriggering (Fig. 4), and
- apnea-hypopneas.
Based on various publications, the percentage of asynchrony affects more than 1 out of 4 patients [15, 16]. The patient ventilator disturbances originate in various sources, including but not limited to bad ventilator performance, inadequate settings and non-intentional leaks. The non-intentional leaks are mostly due to bad mask fitting and have a deleterious effect on ventilator performance [17-23]. The choice of interface is critical to reduce the leaks and assure good ventilation [24]. Meyer et al. wrote about air leaks reducing sleep quality under non-invasive ventilation [25], but specifically the patient ventilator asynchrony that was caused by leaks was not considered. In recent times, patient ventilator asynchrony is being published as having a negative effect on sleep quality for patients using non-invasive ventilation [26, 27].
Manufacturers and clinical or university centres are performing studies to compare or to test ventilators in different conditions and thereby to assess in which conditions patient ventilator disordered breathings occurs.
Bench evaluations of mechanical ventilators are a comfortable way of assessing performance of devices in terms of triggering, cycling, monitoring and leakage behaviour [22, 28-36]. Indeed, technology also brought new developments in the bench design.
However, there are both many patient-specific criteria as well a variety of pathologies that makes this task self-limiting, as can be drawn from the bench evaluation designed for paediatric evaluation by the team of Fauroux [28]. Despite this limitation, bench testing allows repetitive evaluations under similar conditions which is not possible on patients. Indeed, comparison of ventilators on patients demands for a large population to be included with common pathology features to yield a statistically significant analysis else outcomes might be non-significant [37].
As written by Stroetz in 1998, “Practitioners willing to make the effort to interpret patient-ventilator interactions will find it difficult to manage ventilator-dependent patients without feedback from waveform analysis in the future”[38].
This was illustrated by example in a case report by Haynes [39]: by using the waveform display on a Pressure Support ventilator, he was able to detect asynchrony and manipulate inspiratory risetime and breath-termination criteria to improve the patient ventilator synchrony.
Even though use of waveforms to reduce patient ventilator respiratory disturbance has become common in the Intensive Care setting [40-43] and sub-acute/monitoring centres, waveforms could sometimes be difficult to interpret for clinicians not routinely using them. Some computerized algorithms exist for the intensive care setting where patients are deemed more under control [44-46]. Alternative automated methods are being developed specifically for non-invasive ventilation. As an example a, entropy based analysis developed in Rouen [47-49] or an algorithm validated in Nava’s team [16].
- The entropy based system is using a novel algorithm calculating phase portraits and entropy (mostly used in chaos techniques). It looks at the variability of ventilation and triggering efforts. This method is post-hoc by design but could still yield very useful information for the caregiver to adjust the settings (Fig. 6).
- Another algorithm detects ineffective triggering as perturbations in the expiratory flow unaccompanied by a mechanical breath (Fig. 7). This real-time algorithm designed to be embedded in a ventilator yields a sensitivity of 91% and a specificity of 97% according to the authors but on a low number of patients and events, respectively 20 and 159.5±87.7 breaths per patients.
Using waveforms and advanced non-embedded asynchrony detection algorithms, is more unlikely to be done in the home environment for the simple reason that the clinician does not have access to all patient’s vital signs [50]. Monitoring patients’ ventilation implies indeed questioning, examining and monitoring vital signs in addition to looking at the synchronicity of the breathing. The monitoring need is to be correlated with patient’s pathology when selecting a ventilator for use at the home.
Those algorithms and ongoing development have made the Mechnical Ventilators become more technological and potentially more complicated to use. By design, the presence of technology should help the clinician and not overcomplicate the usability of the ventilator. Indeed, lack of ease of use might lead to misunderstanding in ventilation modes and settings and as such create a potential for patient ventilator asynchrony especially when the patient is remote.
In a usability study, J. Gonzales-Bermejo asked 13 ICU physicians to assess the ease of use of 11 home ventilators [51]. When compared with a technical person, the physicians performed poorly yielding the conclusion that user-friendliness of home ventilators might be questionable for physicians not daily using the device.
An additional risk for patient ventilator asynchrony and potential lack of usability, is the lack of common terminology for most of the settings of ventilators. Chatburn proposes a classification system to homogenize the various brand names and ventilation modes although more for the intensive care and sub acute setting [52]. In the home care environment, caregivers will be required to pay extra attention to specific ventilation modes to find the best match between patient and ventilator and ventilator settings.
PITFALLS AND CONSTRAINTS IN THE HOME
Treatment of ventilation has to be performed on a continuous basis in the home. As shown by Petitjean and colleagues on previously stabilized restrictive patients, withdrawal of NIV leads to first nocturnal respiratory failure and within days diurnal respiratory failure [53]. They recommend not stopping NIV for more than a day or two.
With the advent and cost reduction of memory techniques, most manufacturers of home respiratory support ventilators have implemented systems in the devices to collect a number of information on the use of the device. This information can go from simple usage time up to detailed waveforms of pressure, flow, leakage and cycling.
Farré and colleagues reported in a survey on 300 patients in the Barcelona area [54] that approximately one out of seven ventilators exhibited significant discrepancy (> 20%) between prescribed and actual values (1/20 with a difference of more than 30%). In addition the study revealed that when tested, alarms were not working in 0.9% power-off, 18.6% disconnection and 5.1% obstruction for all ventilators. Farré’s study enhances the importance of quality control of home mechanical ventilation. So, technology in home ventilation should also prevent settings to be changed from prescribed values.
In a study in the frame of a European Concerted Action on 326 centres and more than 20000 patients, Farré et al. showed that servicing of the ventilators is mostly dependant on external companies with significant variance in service interval, service centre size and prescriber feedback information [55]. This variability in service provision might be a reason for some of the discrepancy found previously.
Especially when the patient is at home, monitoring the patient and ventilator quality becomes more difficult even if experiments have been successful [56] using telemedicine. The use of telemedicine is a possible path for both monitoring and performing quality control.
In addition to viewing patient waveforms, readjusting ventilator performance and settings could be possible in the home or in relatively remote settings [50, 57-59]. Many commercial non-medical devices are available to achieve that function (Fig. 8). They could be interfaced with the existing ventilators to build the bridge between home and hospital. This approach has been tested and confirmed on OSA patients [60, 61], but not yet on mechanically ventilated patients [62]. One would note that even if telecare has become more present in publications, Botsis and colleagues in a review on Telecare for elderly chronic disease patients revealed that yet less than 10% of the studies published were on COPD or respiratory related pathologies [63].
Though there are no technical limitations, as the bridges operate via most available carriers (Ethernet, WiFi, GPRS, Bluetooth), the care provider should yet consider additional aspects:
- Financial aspect: the cost of using these add-on devices versus traditional approached
- Legal aspect of telecare: a recent report of the European Commission better outlines the framework [64]
- Liability aspect: modules added to medical devices are considered to be medical devices and should inherently be safe [CE Marking: Medical Device Directive (MDD) - 93/42/EEC].
MANUFACTURER’S INITIATIVES TO MONITOR AND IMPROVE PATIENT-VENTILATOR INTERACTION
Tobin et al. identified four major areas of possible problems in interaction: the triggering of the ventilator, the phase of inspiration after triggering, the passage from inspiration to expiration, and the end of expiration [65].
The same causes were identified by Schönhofer and he proposed corrective actions [66], mostly aimed at inspiratory and expiratory triggers, inspiratory risetime and respiratory rates. This work by Schönhofer leads to investigate various critical components and reasons for failure of putting a ventilator on a patient (Fig. 9). Schönhofer and co-author conclude that, in addition to mask adjustment, adjusting some of the ventilator settings is also a key action to reduce patient ventilator disturbances.
Manufacturers are putting significant effort in designing new techniques for improving triggering and cycling techniques to reduce patient ventilator asynchronies. One should consider that with regards to the financial impact, most companies have elected to take a black-box algorithm hiding intrinsic details for the caregiver. The intellectual property is more commonly disclosed first in patent application before being tested clinically, validated and published.
To assess manufacturer’s initiatives, the Intellectual Property was examined on the Wordwide Intellectual Property Office [http: //www.wipo.int/pctdb/en/search-adv.jsp]. In recent years, more than 30 patents have been applied for on various aspects of the asynchrony. Below some of them from various manufacturers:
- Energy Trigger Applicant: BREAS; Publication info: US2008283061
- Method and Apparatus For Detecting Ineffective Inspiratory Efforts and Improving Patient-Ventilator Interaction; Applicant: RESMED; Publication info: US2008110461, based on the work performed by Nava and team [16]
- Determining Suitable Ventilator Settings for Patients with Alveolar Hypoventilation During Sleep; Applicant: RESMED; Publication info: AU2002325399
- Adjustment of Ventilator Pressure-Time Profile to Balance Comfort and Effectiveness; Applicant: RESMED LTD; Publication info: AU2003204620
- Methods and apparatus for varying the back-up rate for a ventilator; Applicant: RESMED LTD; Publication info: NZ541914
- Ventilator patient synchronization; Applicant: RESMED; Publication info: AU2002308423
- Patient-ventilator synchronization using dual phase sensors; Applicant: RESMED; Publication info: EP1810708
- Medical ventilator triggering and cycling method and mechanism; Applicant: RESPIRONICS; Publication info: US2005087190
- Method and device for determining the alveolar ventilation (a.v.) and controlling a respirator; Applicant: WEINMANN; Publication info: EP1985326
- Artificial respiration apparatus adjusting method, involves applying increased volume of respiration gas intermittently with respect to respiration volume for assisted respiration, and realizing deep insufflations by adjustment of pressure; Applicant: WEINMANN; Publication info: DE102007033048
- Method and device for controlling a ventilator; Applicant: WEINMANN; Publication info: DE102007011 924
Although these patent applications are in process, they will not per se be integrated in actual ventilators, as patents are also ways of avoiding competition on one’s unique technique.
Manufacturers yet make other treatment oriented information available for the clinician and the caregiver. The availability of this information opens new paths for documenting the therapy and tracking how the patient is using his breathing assistance device. This memory logging of events discloses the entire therapy without the additional costs linked of in-hospital or ambulatory monitoring (Fig. 10).
CONCLUSION
With the advent of reduced and more powerful electronics, medical equipment in general and non-invasive respiratory support devices in particular have been increasingly made available for the patient in his home environment.
Although beneficial for the patient, this prevents the clinician to have direct control on the patient. Commercially available device relying on various communication techniques now technically give the possibility to remotely monitor the patient in his own familiar environment. The legal and financial framework around this telemedicine is however not entirely ready yet.
Progress in technologies also led to an improved handling of patient-device interaction. Indeed, especially in unsupervised non-invasive ventilation, patient ventilator asynchronies are common. As part of an ongoing effort for better patient-ventilator matching, manufacturers are promoting new or improved ventilation modes. Even though these ventilation modes have become more responsive to patient efforts, adversely they might lead to events such as false triggering, auto-triggering, delayed triggering. Developments for an improved clinician’s feedback on asynchrony and adjusting the ventilator are ongoing.
Finally, in order to track patient/ventilator interaction and usage, manufacturers have embedded memory in most modern respiratory devices. This logging is beneficial for the caregiver to a posteriori analyze the treatment and tune the settings to the patient’s need and comfort.
To conclude there is a need for a joint effort of manufacturers and caregivers (clinicians, service centres) to assure that the patient ventilator interaction is optimized to ensure best treatment.


