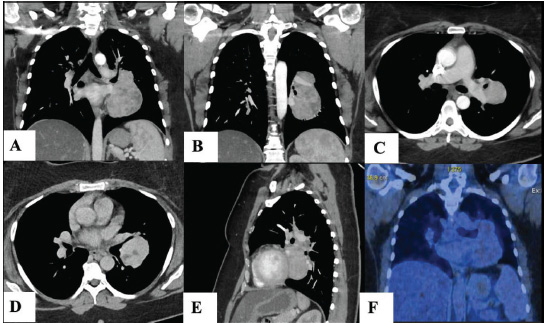
The Open Respiratory Medicine Journal is an open-access journal that publishes research articles, reviews/mini-reviews, letters and guest-edited single-topic issues in all important areas of experimental and clinical research in respiratory medicine. Topics covered include:
- COPD
- Occupational disorders, and the role of allergens and pollutants
- Asthma
- Allergy
- Non-invasive ventilation
- Therapeutic intervention
- Lung cancer
- Lung infections respiratory diseases
- Therapeutic interventions
- Adult and paediatric medicine
- Cell biology
The Open Respiratory Medicine Journal, a peer reviewed journal, is an important and reliable source of current information on important recent developments in the field. Emphasis is placed on publishing quality papers, making them freely available to researchers worldwide.
The Open Respiratory Medicine Journal is an international, peer-reviewed, open-access journal covering all aspects of respiratory medicine published continuously by Bentham Open.