All published articles of this journal are available on ScienceDirect.
Lack of Benefit of High Flow Nasal Oxygen Therapy as Ceiling of Treatment for Severe COVID-19 Pneumonitis in Elderly Frail Patients: A Single Centre Observational Study
Abstract
Background:
Severe COVID-19 pneumonitis in elderly frail patients is associated with poor outcomes, and therefore invasive mechanical ventilation is often deemed an inappropriate course of action. Some evidence suggests high-flow nasal oxygen (HFNO) may prevent the need for invasive ventilation in other groups of patients, but whether it is an appropriate ceiling of care for older frail patients is unknown.
Methods:
We retrospectively identified patients with severe COVID-19 pneumonitis requiring FiO2>60% who were deemed inappropriate for invasive ventilation or non-invasive continuous positive airway pressure ventilation (CPAP). Our local protocol based on national guidance suggested these patients should be considered for HFNO. We observed whether the patients received HFNO or standard oxygen therapy (SOT) and compared mortality and survival time in these groups.
Results:
We identified 81 patients meeting the inclusion criteria. From this group, 24 received HFNO and 57 received SOT. The HFNO group was similar in age, BMI and co-morbidities to the SOT group but less frail, as determined by the Clinical Frailty Scale (CFS). All 24 patients that received HFNO died in comparison to 46 patients (80.7%) in the SOT group. Mortality in the HFNO group was significantly higher than in the SOT group.
Conclusion:
Elderly frail patients with severe COVID-19 pneumonitis deemed inappropriate for invasive ventilation and did not benefit from HFNO. Further, HFNO may have been associated with harm in this group.
1. INTRODUCTION
Severe COVID-19 pneumonitis is associated with poor outcomes in older frail patients as determined by the Clinical Frailty Scale [1]. Advanced age, male gender and significant medical co-morbidities are also associated with worse outcomes [2]. Subsequently, many older frail patients with multiple co-morbidities admitted to the hospital with COVID-19 pneumonitis are deemed inappropriate for invasive mechanical ventilation due to the likelihood of poor outcomes and distress caused by these interventions.
The optimal respiratory support strategy for these patients is unknown. Previous work done in our hospital suggests that this cohort is unlikely to benefit from non-invasive continuous positive airway pressure (CPAP) ventilation [3], although other authors appear to have observed better outcomes in a small retrospective analysis [4]. However, the later study did not include a contemporaneous control group for comparison, and the study included patients with likely, less severe diseases as determined by oxygen requirement at the time of starting CPAP. Additionally, patients often find tightly fitted CPAP masks difficult to tolerate, and alternative strategies are subsequently required. High flow nasal oxygen therapy (HFNO) is considered a potential alternative to CPAP.
HFNO has previously been shown to potentially reduce the need for invasive ventilation in patients with severe hypoxemic respiratory failure [5]. Several recently published observational studies suggest that HFNO may also prevent the need for invasive ventilation in COVID-19 pneumonitis [6-15]. These studies are summarised in a focussed literature review presented in Table 1.
It is, however, important to note that these studies primarily evaluated HFNO in younger patients deemed appropriate for invasive ventilation. They did not address the question of whether HFNO is an appropriate ceiling of treatment for older frail patients. As a result, it is not clear whether a benefit relative to standard oxygen therapy (SOT) would be observed in this patient group. A single pre-print was found examining the potential of HFNO as a ceiling of treatment in frail patients [16]. They observed a 75% mortality in an elderly frail group that as failing to maintain oxygen saturation with 15 L/min O2 via a standard mask with a non-rebreathe reservoir. The authors did not include a comparison group that utilised an alternative treatment strategy. Although the study included older frail individuals, the median age of the eight survivors was 69.5 years.
In our institution, patients with severe COVID-19 pneumonitis deemed inappropriate for invasive ventilation and requiring an inspired concentration of O2 greater than 60% were considered for HFNO. We retrospectively identified all patients meeting these criteria within a four-month period in our institution. The patients were reviewed in order to compare the outcomes of those who received HFNO to those who received SOT.
2. METHODS APPROVALS AND SETTING
After consultation with the chair of the local research ethics committee, no formal ethical approval for this study was sought as this was considered a service evaluation. All interventions were carried out at Kettering General Hospital (United Kingdom, a 600 bedded secondary care hospital serving a population of 330,000).
We prospectively identified all consecutive COVID-19 cases with severe type 1 respiratory failure requiring FiO2 at 0.6 admitted to our hospital between 23 September 2020 and 24 January 2021 that were deemed inappropriate for invasive ventilation or CPAP. Clinicians made decisions regarding the suitability of patients for the trial of HFNO based on CFS. Their outcomes were recorded and compared with a similar group of patients who received standard oxygen as a ceiling of treatment. Patients were followed up until death or discharge.
2.1. Decision for Ceiling of Treatment Plan and Ventilatory Support
A decision on fitness for invasive ventilation, including do not attempt cardiopulmonary resuscitation order (DNACPR), was recorded in the medical notes at the time of admission after senior clinician review and discussion with the patient as per national guidelines. Patients who met the criteria for HFNO but were deemed too ill to benefit from HFNO due to baseline frailty and the poor likelihood of tolerance to ventilatory devices were included in the SOT group.
2.2. Patient Selection and Classification
We retrospectively identified all COVID-19 (confirmed) patients with severe respiratory failure who required FiO2 at 0.6 and were admitted to our hospital between 23 September 2020 and 24 January 2021. All confirmed cases had a positive RT-PCR nasopharyngeal swab for COVID-19. Patients requiring NIV for acute or chronic type 2 respiratory failure due to pre-existing conditions were excluded from the study. The 4C Mortality Score for COVID-19 was calculated from recorded patient parameters, including age, gender, number of co-morbidities, respiratory rate, peripheral oxygen saturation, Glasgow coma scale, urea level and C-reactive protein. The 4C Mortality Score [17] was used as a risk stratification score for mortality on the day of admission (retrospectively).
2.3. Inclusion Criteria
- Age 18 and above.
- RT-PCR confirmed for COVID-19.
- Requiring FiO2 of 0.6 or more to maintain SpO2 >92% (88-92% in COPD).
- Not fit for invasive ventilation with DNACPR in place, based on Clinical Frailty Score (CFS) and existing guidelines. Decision recorded on admission, prior to treatment.
2.4. Exclusion Criteria
- Patients appropriate for Intensive care unit level escalation / invasive ventilation / CPAP.
- Patients requiring CPAP or BiPAP for ‘acute’ or ‘acute on chronic’ type 2 respiratory failure.
2.5. Delivery of HFNO and SOT
In the HFNO group, we used Fisher & Paykel Healthcare’s Airvo™ 2, a humidified Nasal High Flow system, to deliver respiratory support for patients who were fit for HFNO. The system provides an FiO2 of up to 98% depending on the flow rate of oxygen, up to a maximum flow rate of 60 litres per minute. The flow rate of oxygen was used to titrate the FiO2, whilst the total flow rate was held at 60 litres per minute. Using this method, the FiO2 was titrated and recorded to achieve target saturations for the patient as measured by pulse oximetry (92-96%, or 88-92% for those at risk of type 2 respiratory failure). All patients on HFNO had continuous pulse oximetry monitoring in high visibility areas of the hospital and received immediate review of FiO2 if saturations dropped below the target range. Arterial blood gases were not routinely performed for titration of oxygen. If patients were unable to maintain acceptable saturations on 98% FiO2, and if no other reversible cause could be found and treated, patients received personalised symptomatic end-of-life care.
In the SOT group, the flow rate of oxygen was a maximum of 30 litres/minute via double humidifier oxygen or 15 litres/minute delivered via a venturi mask or humidified circuit. When patients could not maintain target saturations with this therapy, a non-rebreathing bag was used with a flow rate of 15 litres/minute.
2.7. Focussed Literature Review
A PubMed database search for all studies of HFNO in COVID-19 pneumonitis was conducted using the following search terms: COVID-19; COVID-2019; SARS-CoV-2; high flow nasal oxygen; high flow nasal cannula; pneumonia; ventilation.
2.8. Statistical Analyses
As this study was proposed as a service evaluation, a power calculation was not conducted. Continuous data were described with a mean and associated standard error of the mean (SEM) or median and interquartile range (25th – 75th percentile) where appropriate. Continuous variables were compared by utilising the Student’s unpaired t-test. Categorical data were expressed as a frequency and percentage with a comparison between groups conducted with the χ2 test . The proportion of patients surviving in each group over time in days since admission was plotted, and the survival curves generated for each group were compared by the Mantel-Cox test. A P<0.05 was considered statistically significant.
All data were analysed using GraphPad Prism software version 7.04 (GraphPad Software, Inc, CA, US).
3. RESULTS
Eighty-one patients were included following compliance with the aforementioned inclusion criteria and exclusion criteria. Twenty-four and fifty-seven patients were allocated to the HFNO and SOT groups, respectively. The groups did not exhibit a statistically significant difference in terms of age, gender, BMI, or any of several co-morbidities identified as being strongly associated with mortality in the OpenSafely study [2] (Table 2). The HFNO group had a significantly lower Clinical Frailty Scale score (Table 2).
| Study | Country/ Setting | Type of Study | HFNO/n | Age/years | Male (%) | Co-morbidities (%) | Successfully Weaned off (%) | Escalation to NIV/IV (%) | Death (%) | ||
|---|---|---|---|---|---|---|---|---|---|---|---|
| HTN | DM | IHD | |||||||||
| (Wang, Zhao et al. 2020)[15] | China/ N/A* | Retrospective observational study | 17 | 65 (56-75) | 41 | 18 | 18 | 18 | 10 (59) | 7 (41.2) | N/A* |
| (Calligaro, Lalla et al. 2020)[14] | South Africa/ICU (36%) +non ICU (64%) | Prospective observational study | 293 | 52 (44-58) | 56 | 45 | 54 | N/A* | 137 (46.8) | 111 (37.9) | 45 (15.4) |
| (Yang, Yu et al. 2020)[13] | China/ICU (100%) | Retrospective observational study | 33 | N/A* | N/A* | N/A* | N/A* | N/A* | 17 (51.5) | N/A* | 16 (48.5) |
| (Guy, Créac'Hcadec et al. 2020)[12] | France/Pulmonology ward (100%) | Prospective observational study | 27 | 77 (77-79) | 81 | N/A* | N/A* | N/A* | 19 (70.4) | 1 (3.70) | 4** (14.8) |
| (Zhou, Yu et al. 2020)[11] | China/ N/A* | Retrospective cohort study | 41 | N/A* | N/A* | N/A* | N/A* | N/A* | 8 (19.5) | N/A* | 33 (80.5) |
| (Luo, Xia et al. 2020)[10] | China/ N/A* | Retrospective observational study | 106 | N/A* | N/A* | N/A* | N/A* | N/A* | 32 (30.2) | N/A* | 74 (69.8) |
| (Liao, Chen et al. 2020)[9] | China/ N/A* | Multi-centre prospective observational study | 31 | N/A* | N/A* | N/A* | N/A* | N/A* | 18 (58.1) | *** | 6 (19.4) |
| (Hu, Zhou et al. 2020)[8] | China/Respiratory ward (100%) | Retrospective cohort study | 105 | 64 +/- 11.3 | 48.6 | 57.1**** | 65 (61.9) | 24 (22.9) | 16 (15.2) | ||
| (Demoule, Vieillard Baron et al. 2020)[7] | France/ICU (100%) | Prospective observational study | 146 | 60 (53-67) | 79 | 46 | 29 | N/A* | 64 (43.8) | 82 (56.2) | 30 (20.5) |
| (Katsuno, Suzuki et al. 2021)[6] | Japan/ N/A* | Retrospective observational study | 15 | 68 (64-77) | 80 | 73.3 | 60 | 20 | 8 (53.3) | 6 (40) | 3***** (20) |
**-The outcome of 4 patients were unknown as they were still on HFNC therapy at the end point of the study
***-7 patients were reported under prolonged recovery with eventual outcome not reported
****-The study reported the pooled prevalence of co-morbidities as opposed to prevalence of specified co-morbidities
*****-Two of the patients died following a period of invasive ventilation
HTN Hypertension, DM Diabetes mellitus, IHD Ischaemic heart disease, ICU Intensive care unit
| Patient Demographics | HFNO (n=24) | SOT (n=57) | P value |
|---|---|---|---|
| Gender | |||
| Male (%) | 62.5 | 54.4 | 0.501 |
| Age/years | 79.9 +/- 2.01 | 79.7 +/- 1.31 | 0.97 |
| BMI/ kg/m2 | 28.0 +/- 2.15 | 29.0 +/- 1.26 | 0.705 |
| CFS (IQR) | 4 (4-6) | 6 (5-6) | 0.006 |
| 4C Mortality Score for COVID-19 | 14 (12.25-15) | 14(12-15) | 0.994 |
| Co-morbidities (%) | |||
| HTN | 45.8 | 40.4 | 0.906 |
| DM | 33.3 | 38.3 | 0.461 |
| Cardiovascular | 25 | 14.9 | 0.442 |
| Chronic respiratory disease | 29.2 | 17.0 | 0.545 |
| Neurological impairment* | 20.8 | 55.3 | 0.07 |
| Asthma | 12.5 | 4.26 | 0.423 |
| Malignancy | 16.7 | 4.26 | 0.095 |
| Chronic Kidney Disease | |||
| eGFR<60 mL/min/1.73m2 | 20.8 | 23.4 | 0.877 |
| eGFR<30 mL/min/1.73m2 | 12.5 | 8.51 | 0.423 |
| Autoimmune | 8.33 | 2.13 | 0.360 |
| Patient Demographics | HFNO (N=24) | SOT (N=57) | Relative Risk (95% CI) |
|---|---|---|---|
| - | |||
| Interval from admission/days | - | - | - |
| Initiation of FiO2 60% oxygen therapy | 2 +/- 0.576 | 2 +/- 0.51 | N/A |
| Initiation of FiO2 > 60% oxygen therapy | 2 +/- 0.566 | 3 +/- 0.57 | N/A |
| Initiation of HFNO oxygen therapy | 3 +/- 0.649 | N/A* | N/A |
| Mortality | - | - | - |
| Inpatient death (%) | 24 (100) | 46 (80.7) | 1.20 (1.02 – 1.33) |
| Interval from admission to mortality/days | 8 +/- 1.68 | 9.5 (1.43) | N/A |
| Patient Demographics | Survivors (11) | Non-survivors (n=46) | P value |
|---|---|---|---|
| Gender | |||
| Male (%) | 63.6 | 52.2 | 0.493 |
| Age/years | 70.8 +/- 6.07 | 80.8 +/- 1.53 | 0.0226 |
| BMI/ kg/m2 | 30.4 +/- 2.56 | 28.1 +/- 1.51 | 0.434 |
| CFS | 5 (4.5-6) | 6 (5-6) | 0.248 |
| 4C Mortality Score for COVID-19 | 14 (12-16) | 14 (12-15) | 0.751 |
| Co-morbidities (%) | |||
| HTN | 45.5 | 42.1 | 0.802 |
| DM | 54.5 | 42.1 | 0.352 |
| Cardiovascular | 27.3 | 17.5 | 0.345 |
| Chronic respiratory disease | 45.5 | 22.8 | 0.0463 |
| Neurological impairment* | 18.2 | 47.4 | 0.0223 |
| Asthma | 18.2 | 7.02 | 0.0954 |
| Malignancy | 9.09 | 5.26 | 0.527 |
| Chronic Kidney Disease | |||
| eGFR<60 mL/min/1.73m2 | 27.3 | 24.6 | 0.816 |
| eGFR<30 mL/min/1.73m2 | 0 | 7.02 | 0.311 |
| Autoimmune | 9.09 | 3.51 | 0.255 |
| Interval from admission/days | |||
| Initiation of FiO2 60% oxygen therapy | 1 (0-2.5) | 3 (1-4.75) | 0.0978 |
| Initiation of FiO2 > 60% oxygen therapy | 1 (0.5-3) | 3 (1.25-5) | 0.157 |
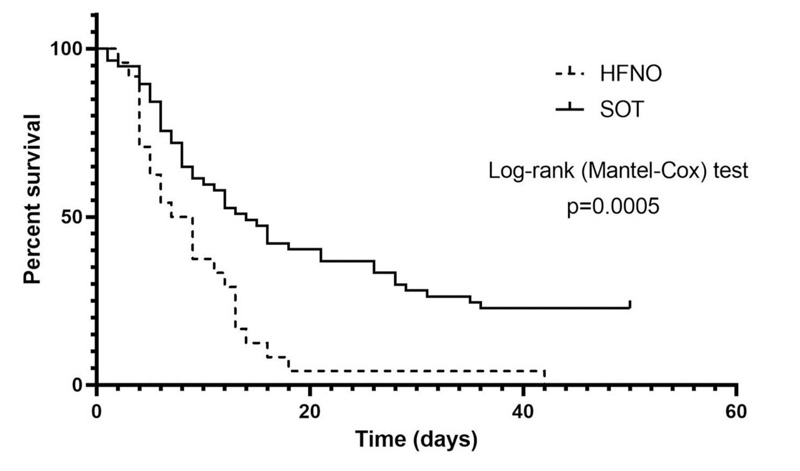
The rate of deterioration of both groups from admission to needing 60% oxygen therapy was similar (Table 3). The SOT group was started on FiO2 > 60% at around the same time as when the HFNO group commenced HFNO therapy. All 24 patients who received HFNO therapy died. Forty-six patients (80.7%) in the SOT group died and the remaining patients survived until hospital discharge. The primary outcome of mortality was significantly worse in the HFNO group (Table 3). All survivors were in the SOT group and a comparison of survivors to non-survivors within this group showed that they were younger with different co-morbidity profiles (Table 4). Survival time analysis of up to 50 days similarly revealed shorter survival in the HFNO group (Fig. 1).
4. DISCUSSION
The data obtained in this study suggest that HFNO offered no benefit to older frail patients with severe COVID-19 requiring FiO2 > 60% and deemed inappropriate for invasive ventilation. All patients that received-HFNO died, while 11 out of a total of 57 patients survived to discharge in the SOT group. This result suggests the possibility of potential harm from HFNO in this group.
This study has some important limitations that need to be considered. The total HFNO sample size was less than initially desired, with 24 patients included in this single centre study. This was the case because once poor outcomes became clear to the clinical decision-makers, they were less inclined to consider HFNO therapy. This was an observational study, and therefore our findings do not exclude other possible interpretations of the data. The groups were very similar ages (mean age 79.9 years [HFNO] vs 79.7 years [SOT]), and although the HFNO group had a higher proportion of males (62.5% vs 54.4%), this was not a significant difference. We, however, acknowledge the discrepancies in baseline demographics between the two groups. The standard oxygen therapy (SOT) group demonstrated a higher clinical frailty score (CFS) compared to the high flow nasal oxygen (HFNO) group. This was because the SOT patient cohort was initially reviewed by a respiratory physician and subsequently considered inappropriate and unlikely to benefit from HFNO therapy due to reasons such as severe frailty. As a result, they were instead treated with SOT.
Relative to other-data available in published literature, this study has the strength of including an imperfect but meaningful comparison group.
However, it should be noted that the Clinical Frailty Scale score of those who received HFNO was, on average, two points lower in comparison to the SOT group. As stated above, a higher score on this scale has previously been shown to be associated with worse outcomes in a large observational study [1]. The rate of deterioration from hospital admission until needing FiO2 60% to maintain O2 saturations was very similar in both groups, suggesting that the severity of respiratory failure was similar in both groups. However, this does not exclude the possibility that the HFNO group may have been deteriorating more rapidly after this point.
Despite these limitations, the observations in this study are stark. It suggests that future studies for the use of HFNO in severe COVID-19 pneumonitis should be closely monitored for safety signals, particularly in the elderly frail cohort. The lack of benefit is consistent with the results of the RECOVERY-RS study preprint [18]. The RECOVERY-RS is a multi-centre, three-arm randomised control trial comparing CPAP, HFNO or conventional oxygen therapy with the primary outcome presented as a composite of invasive mechanical ventilation or mortality within 30 days. No significant difference was observed when comparing HFNO with conventional oxygen. Our study findings, however, contrast with the findings presented by van Steenkiste et al. in pre-print [16]. Van Steenkiste et al. observed 25% survival when HFNO was used as a last resort in elderly frail patients. However, the median age of their survivors was 69.5 years and a meaningful comparison group was not included/presented. Therefore, conclusions regarding the benefit of HFNO over SOT cannot be made from their study. The mean age of survivors in our study (all in the SOT group) was similar (70.8 +/- 6.07 years). Further, it may be pertinent to note that in the van Steenkiste study HFNO was utilised when patients had developed a higher O2 requirement. While the findings of the RECOVERY-RS trial have guided current clinical practise, it is important to consider the difference in demographics between our study and the population recruited for the trial. The patients recruited for RECOVERY-RS were not at their ceiling of care and considered appropriate for escalation to invasive mechanical ventilation. As a result, we believe further RCTs are necessary to assess suitable treatments options for patients currently at ward-based ceiling of care and unsuitable for critical care intervention.
There are several potential mechanisms by which we may hypothesise that HFNO may be associated with harm. The nasal mucosa appears to be the site with the highest viral burden, thus, aspiration-mediated seeding virus to the lung is a plausible explanation for the distribution of viral infection and pneumonitis observed [19]. It may be possible to suggest that HFNO has a role in the facilitation of aspiration and seeding. Furthermore, relatively higher concentrations of O2 may exacerbate organ injury via alternative mechanisms that have been observed in other conditions [20].
Additionally, the need to wear high-level personal protective equipment when caring for elderly frail patients receiving HFNO (deemed an aerosol-generating high risk procedure) may compromise or affect the quality of their nursing care (in comparison to those receiving SOT that were cared for by staff using lower-level personal protective equipment). However, patients requiring HFNO had a lower ratio of patients to nursing staff to compensate for these issues, i.e., nurses cared for a maximum of 4 patients receiving HFNO vs. 7 patients receiving SOT.
CONCLUSION
In summary, our study does not show any evidence of survival advantage from HFNO treatment in COVID-19 pneumonitis when compared to conventional oxygen therapy amongst patients who were unsuitable for invasive mechanical ventilation or CPAP. In fact, findings from the study even suggest potential harm from HFNO in this particular population group.
While HFNO facilitation in the ward setting has been demonstrated to be feasible, this study has conveyed some important points for consideration with respect to treatment efficacy and mortality. As a result, current treatment strategies for this patient population may require further reflection and evaluation in order to develop robust treatment strategies.
LIST OF ABBREVIATIONS
| HFNO | = High-Flow Nasal Oxygen |
| CPAP | = Continuous Positive Airway Pressure Ventilation |
| CFS | = Clinical Frailty Scale |
ETHICAL STATEMENT
After consultation with the chair of the local research ethics committee, no formal ethical approval for this study was sought as this was considered a service evaluation.
CONSENT FOR PUBLICATION
Informed consent was obtained.
STANDARD OF REPORTING
STROBE guidelines were followed.
AVAILABILITY OF DATA AND MATERIAL
The authors confirm that the data supporting the findings of this study are available within the article.
FUNDING
None.
CONFLICT OF INTEREST
The authors have no conflicts of interest to declare.
ACKNOWLEDGEMENTS
Declared none.


