All published articles of this journal are available on ScienceDirect.
Acute Pericarditis as the Primary Presentation of COVID-19 Infection followed by Guillain-Barre Syndrome in a Healthy Young Man: A Case Report
Abstract
Background:
Symptomatic COVID-19 infection most often presents as acute respiratory distress syndrome. Acute pericarditis and Guillain–Barré syndrome are rare extrapulmonary manifestations of this infection.
Case Presentation:
A 27-year-old man presented with chest pain, with negative troponin and typical electrocardiographic findings, resulting in a diagnosis of acute pericarditis. He had no respiratory symptoms, nor the chest computerized tomography (CT) scan findings of COVID-19, and his Polymerase chain reaction (PCR) was negative. One week later, he developed clinical symptoms of Guillain-Barre syndrome, along with respiratory manifestations of COVID-19. His repeat chest CT scan and PCR test confirmed COVID-19 infection. After 17 days of hospital stay, he improved clinically and was discharged.
Conclusion:
This is the first case of acute pericarditis as the primary presentation of COVID-19 in the absence of respiratory symptoms and a clear chest CT scan, followed by the development of Guillain–Barré syndrome and respiratory tract manifestations of COVID-19. Clinicians should be aware of the extrapulmonary presentation of COVID-19 infection.
1. INTRODUCTION
Coronavirus Disease 2019 (COVID-19) emerged in November, 2019, in Wuhan, China, and due to its extremely contagious nature, it soon resulted in a worldwide viral pandemic. An acute respiratory syndrome is the typical presentation of the disease, resulting from viral proliferation, which causes cytokine release, capillary inflammation, fibrin disposition, and mucositis [1-3]. COVID-19 most commonly presents as a respiratory infection manifested by cough, dyspnea, and acute respiratory distress syndrome (ARDS) [4, 5].
Although the respiratory system is usually the main focus of COVID-19 infection, inflammation of other organs is also possible. An exaggerated immune response is considered responsible for the various presentations simulating other autoimmune inflammatory diseases. The key mechanism by which SARS-CoV-2 establishes its multiorgan effects is through virus-induced immune response, which mostly results from hyperinflammation, toxicity of the virus, endothelial cell injury, and renin-angiotensin-aldosterone system (RAAS) dysregulation [6-9]. Patients’ age, sex, and race influence the presentation and severity of the disease; the exact pathogenesis of these presentations is not completely known. The mechanism is explained by the term ‘cytokine storm’, defined as immune system dysregulation through activation of systemic inflammation by cytokine release, leading to organ failure [10].
COVID-19 can lead to a variety of cardiac presentations, such as myocardial ischemia, myocarditis, arrhythmias (new-onset atrial fibrillation and flutter), cardiomyopathy, cardiac failure and cardiogenic shock, as well as thromboembolic events [9, 11]. There is a growing trend in neurological problems of COVID-19 infection. COVID-19 is reported to result in numerous neurological manifestations, either by causing direct neuronal injury, e.g., encephalitis, encephalopathy, and cerebrovascular events or via neuronal inflammation like Guillain–Barré syndrome (GBS) and acute myelitis [9, 10, 12, 13]. In this study, we represent a rare case of COVID-19 in a 27-year-old healthy man with a primary presentation of acute pericarditis, followed by Guillain-Barre syndrome.
2. CASE PRESENTATION
A 27-year-old formerly healthy athlete male was presented to the emergency department with a chief complaint of acute-onset pressure-like pleuritic chest pain. His chest pain started in the morning of the presentation, worsened gradually, and was aggravated by lying supine. He denied any history of fever, cough, sputum production, shortness of breath, myalgia, arthralgia, diarrhea, or headache in the preceding days. On physical examination, he was hemodynamically stable, not dyspneic but preferred leaning forward. His heart rate was 80 bpm with blood pressure of 110/60 mm Hg, respiratory rate of 18 breaths/min, O2 saturation of 95% on room air, and temperature of 36.7. Physical examination, including the cardiovascular system, was unremarkable, with no pericardial friction rub.
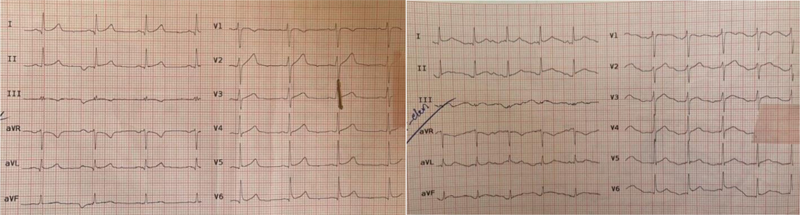
The primary laboratory results were white blood cell count of 9000/mm3 with 25% lymphocytes, platelet count of 165000, ESR: 7 mm/hour, CRP: 10 mg/L, and D-dimer: 350 ng/ml. The liver and kidney function tests were normal. His creatine phosphokinase (CPK) was 1013 U/L, and cardiac troponin T (hs-cTnT) on admission and at 6 hours was 0.04 ng/mL and 0.03 ng/mL, respectively. The electrocardiogram (ECG) showed a slight diffuse ST elevation in most leads, a reciprocal ST depression, and PR segment elevation in AVR, with PR segment depression in other leads (Fig. 1). Chest radiograph showed no pathology. Chest Computerized Tomography (CT) scan and Computerized Tomography Angiography (CTA) revealed no lung involvement, pneumothorax, pericardial effusion, or pulmonary embolism. Transthoracic echocardiogram showed normal structure and function, with no regional wall motion abnormalities and no pericardial effusion. Based on European Society of Cardiology (ESC) guidelines, acute pericarditis is diagnosed in the company of two of the following four criteria: pleuritic chest pain, pericardial friction rub, ECG findings suggestive of pericarditis, pericardial effusion [14, 15]. Clinical judgment of acute pericarditis in our patient was based on his clinical presentation (pleuritic chest pain) and typical ECG findings. Due to the COVID-19 pandemic situation, despite the inconclusive report of the chest CT scan, samples of nasopharyngeal swabs were sent for SARS-CoV-2 RT-PCR testing, which was reported as negative. The complete set of rheumatologic, endocrine, and serological viral marker tests was within the normal range. The patient was admitted to CCU and received 8-hourly Ibuprofen (400mg) and Aspirin (325mg), as well as 4 L/min oxygen supplement via mask.
On the 9th day of admission, the patient developed dyspnea and fever of 38.1. His oxygen saturation decreased to 86% on room air, and his respiratory rate increased to 24 breaths/min, with a blood pressure of 100/65 and a heart rate of 98 bpm. His lung auscultation revealed coarse breath sounds and coarse crackles bilaterally. A repeated chest CT scan revealed bilateral multi-lobar consolidation with peripheral distribution, suggestive of COVID-19 infection (Fig. 2). A repeat nasopharyngeal swab was sent for SARS-CoV-2, which came back positive. Concurrently, he reported symptoms of acute and progressive bilateral distal extremity weakness, predominantly involving the left lower limb. On neurologic examination, the sensation was reduced in both lower limbs, and muscle power was decreased to 2/5 in the left distal lower limb, 3/5 in the right lower limb, and 4/5 in both upper limbs. Deep tendon reflexes were reduced in both the upper and lower extremities. He had difficulty walking with an unsteady gait; his plantar reflexes were normal. No urinary retention nor spinal sensory level was noted. Repeated laboratory examination revealed leukopenia and lymphopenia (WBC count of 2900/mm3 with 15% lymphocytes) as well as thrombocytopenia (platelet count of 95000). Other lab tests were as follows: CRP: 80mg/L, LDH: 455 U/L, Ferritin: 460 ng/mL, CPK: 174 U/L, and elevated D-Dimer level of 250 ng/ml. Other laboratory findings were unremarkable.

An Electromyography/Nerve Conduction Study (EMG/ NCS) revealed decreased amplitude of tibial and peroneal sensory and motor action potentials. Both latencies and nerve conduction velocity were normal, as were the F-wave and H-reflexes. A nerve conduction study in the upper limbs showed no abnormalities. EMG study of both upper and lower limb muscles showed mild denervation potential in lower limb muscles and the distal muscles of the upper limbs. These findings revealed an acute, mild, mainly axonal peripheral polyneuropathy, with symmetric sensorimotor involvement, suggestive of a subtype of Guillain-Barre syndrome (GBS), namely the Acute Motor and Sensory Axonal Neuropathy (AMSAN). The cerebrospinal fluid (CSF) examination was performed on the 7th day of hospitalization and showed a white blood cell count of 4/ml and protein of 50 mg/dl. The whole spine MRI reported insignificant findings. The diagnosis of AMSAN was made in accordance with the Brighton Collaboration GBS Working Group criteria, based on the presence of bilaterally reduced lower extremity muscle strength and findings of motor and sensory damage on his Nerve Conduction Studies (NCS) [16].
The patient was transferred to COVID-19 ICU and received 10 L/min of saturated oxygen via a non-rebreather mask. Remdesivir 200mg was administered on day 1, followed by a 100mg daily dose for a further 9 days. He was given Enoxaparin (40 mg SC daily), Hydroxychloroquine (400 mg on day one, followed by 200 mg BD), and dexamethasone 8 mg BD for 10 days. Other treatments included: Synairgen: Interferon-beta-1a (44 units SC BD), and intravenous immunoglobulin 0.4 g/kg/day for 5 days. By the 21st day of the hospital stay, the patient had significant improvement in his clinical state, including his motor deficit and was discharged home.
3. DISCUSSION
Several cardiovascular complications have been reported with COVID-19 infection, leading to more probability of morbidity and mortality in patients affected [11]. Of these complications, conditions, such as MI, myocarditis, cardiac failure, arrhythmia, and cardiogenic shock, are the more commonly reported ones, while disorders, such as acute pericarditis, acute myopericarditis, large pericardial effusion, and lethal tamponade, are less common [17-24]. Based on autopsy examinations, pericarditis rates of 19-22% have been reported in COVID-19 patients [25, 26]. Pericardial involvement stems from pericardial inflammation, which is the result of the cytotoxic effects of T and B cell proliferation by the virus and activation of the immune system [14, 27]. A definite etiology of pericardial involvement in COVID-19 is not completely known [28]. Postulated mechanisms for inflammation in acute pericarditis include systemic cytokine release, endothelial dysfunction, and T-cell-mediated immunopathology [28, 29].
A literature review of case reports of acute pericarditis in COVID-19 infection from the beginning of the pandemic until July, 2021, resulted in 35 studies, including 38 cases. These cases are divided into four groups. The first group (24 cases) developed pericarditis following respiratory manifestations, most of whom had pulmonary involvement on chest CT scans at the time of admission [15, 18-20, 23, 30-46]. In the second group (7 cases), acute pericarditis was the main presentation of COVID-19 in the development of the disease. These patients presented with mild respiratory symptoms, but their chest CT scan was clear [17, 24, 47-51]. The third group included 3 case reports of acute pericarditis in patients presenting with nonrespiratory manifestations of COVID-19, such as diarrhea, vomiting, and chest pain, who had lung involvement on a chest CT scan at the first visit [52-54]. The fourth group included acute pericarditis as the primary presentation of COVID-19 in the absence of respiratory manifestations and lung involvement on chest CT scan; this has been observed in 3 adults and 2 adolescents with COVID-19 [22, 55-57] (Table 1). The present study is about a very rare case of acute pericarditis as the primary presentation of COVID-19 infection, who later, during the disease and concomitant with the occurrence of respiratory manifestations, developed another infrequent extrapulmonary complication, the Guillain-Barre syndrome.
| Case/Article | Patient’s Age/Sex | Interval between Primary Presentation and Respiratory Symptom Development | Pericardial Effusion (PE), Diagnosed by TTE | PCR at the Time of Admission | Outcome |
|---|---|---|---|---|---|
| Current case | 27 yr old Male |
1 week (Dyspnea, fever Hypoxia) |
No PE | Negative, Repeated PCR positive (day 7) |
Discharged on the 17th day of admission |
| Osman et al. [22] |
50 yr old Male | Day 5 (Dyspnea, dry cough, fever) |
Moderate PE | Positive | Discharged |
| Kumar et al. [55] |
66 yr old Male |
Day 6 (Dry cough, fever) |
Negative for PE but echo bright, mildly thickened pericardium |
Negative, Repeated PCR positive (day 6) |
Discharged, readmitted on day 6, discharged on day 12 |
| Naqvi et al. [56] |
55 yr old Male |
No respiratory symptoms | Mild to moderate PE | Positive | Discharged |
| Dimopoulou et al. [57] |
14.5 yr old Male |
No respiratory symptoms | Mild to moderate PE | Negative, Repeated PCR positive (day...) |
Discharged on day 4th day of admission |
Of the five COVID-19 cases primarily presenting with acute pericarditis, none had lung involvement on a chest CT scan at the time of admission. None of the cases developed tamponade or needed mechanical ventilation. All cases improved gradually, and they were discharged from the hospital.
Recent studies have reported the occurrence of autoimmune neurological complications, such as acute immune-mediated polyneuropathy or GBS, following SARS-CoV-2 [58, 59]. Several cases were reported of patients with COVID-19 infection developing a variety of neurological manifestations [12, 60-62]. Guillain-Barre syndrome is an uncommon condition, presenting with muscle weakness and paresthesia from peripheral nerve demyelination and neuronal damage. It is speculated that the mechanism of neuronal injury in GBS involves an immune-mediated response, which results in cross-reactivity of infection with elements of peripheral nerves [63]. GBS has been observed in the setting of various infections, for instance, Campylobacter jejuni, Flaviviruses, cytomegalovirus, Mycoplasma pneumonia, Epstein-Barr virus, and Orthomyxoviruses [64-67]. The precise pathophysiology of GBS in COVID-19 infection is unclear. Neurotropism and immune intermediated damage are possible theories, although RT-PCR of CSF failed to detect SARS-CoV-2 [60, 68]. AMSAN is a form of acute motor axonal neuropathy (AMAN), a variant of GBS, which mostly affects young adults, particularly in Asia [69, 70]. Diagnosing the condition is based on the presence of an axonal loss in the absence of nerve demyelination and is suggested by the reduced amplitude of compound muscle action potential (CMAP) and sensory nerve action potentials (SNAP) [71].
Several GBS cases have been described in COVID-19-affected cases since the outbreak of the infection in Wuhan, China [58-60, 72-74], including a case of a pregnant woman reported by Mehrpour et al. [75]. A systematic review by Sheikh et al. evaluated 64 studies with a total of 94 GBS cases in COVID-19-infected patients and concluded that respiratory manifestations were the most frequent primary presentation in this population (72.35%); fever and GI symptoms were the initial presenting features in 65.95%, and 19.15% of the patients, respectively. A significant overlap of presenting symptoms was observed. Paresthesia (48.93%), paresis of the lower extremity (41.49%), and upper extremities (22.34%) were the most common neurological findings. No situation of GBS in COVID-19 was stated with the initial presentation of acute pericarditis. Of the 83 patients in whom the GBS subtype was mentioned, 70% had Acute Inflammatory Demyelinating Polyneuropathy (AIDP), 12% had AMSAN, 12% were diagnosed with the Miller Fisher syndrome, and 6% had AMAN. RT-PCR from the nasopharyngeal samples was positive in 69.15% of patients [76].
In the present case, initial COVID-19 RT-PCR testing on the nasopharyngeal samples was negative at the time of presentation with acute pericarditis. Acute pericarditis was the primary presentation in the absence of respiratory symptoms and chest CT scan findings. The course of the disease was later complicated by GBS, the occurrence of which was concomitant with the emergence of respiratory tract manifestations. After 1 week of therapy with Ibuprofen and high-dose Aspirin for acute pericarditis, the neurologic symptoms of GBS became evident, and chest CT revealed changes suggestive of COVID-19 infection, which was confirmed after COVID-19 RT-PCR on a nasopharyngeal sample that came back positive. To the finest of our knowledge, this is the first rare case of acute pericarditis as the primary presentation of COVID-19, which gets complicated by another rare extrapulmonary manifestation of COVID-19, GBS. The occurrence of two extrapulmonary organ involvement in the present case may have an autoimmune inflammatory etiology caused by cytokine storm, which is a frequent state of COVID-19 infection. No specific recommendations exist with regard to the treatment of acute pericarditis in the setting of COVID-19 [28]. Up until now, there has been no contradiction in the usage of high-dose aspirin in COVID-19 patients [77] rather, early initiation of NSAIDs, including ibuprofen, has been proposed to be helpful in the prevention of COVID-19 complications [78].
CONCLUSION
We present a case of pericarditis as the primary presentation of COVID in the absence of respiratory involvement, followed by symptoms consistent with Guillain-Barre and respiratory symptoms at the time of confirmed COVID-19 infection. To get a valid result of optimization, this would be critical for clinicians to consider extrapulmonary presentations of COVID-19 infection, including cardiac and neurologic involvement, together or separately, such as in this case of concurrent acute pericarditis and Guillain–Barré syndrome.
AUTHORS’ CONTRIBUTIONS
All authors read and approved the final manuscript. All authors take responsibility for the integrity of the data and accuracy of the data presented.
LIST OF ABBREVIATIONS
| COVID-19 | = Coronavirus Disease 2019 |
| CT | = Computerized Tomography |
| PCR | = Polymerase Chain Reaction |
| AMAN | = Acute Motor Axonal Neuropathy |
| CMAP | = Compound Muscle Action Potential |
| SNAP | = Sensory Nerve Action Potentials |
| RT-PCR | = Real-Time Reverse-Transcriptase Polymerase Chain Reaction |
| CSF | = Cerebrospinal Fluid |
| SARS-CoV-2 | = Severe Acute Respiratory Syndrome Coronavirus 2 |
| ARDS | = Acute Respiratory Distress Syndrome |
| RAAS | = Renin Angiotensin–Aldosterone System |
| GBS | = Guillain Barré Syndrome |
| CPK | = Creatine Phosphokinase |
| hs-cTnT | = Cardiac Troponin T |
| ECG | = Electrocardiogram |
| CTA | = Computerized Tomography Angiography |
| ESC | = European Society of Cardiology |
| EMG/NCS | = Electromyography/Nerve Conduction Study |
ETHICS APPROVAL AND CONSENT TO PARTICIPATE
Not applicable.
HUMAN AND ANIMAL RIGHTS
Not applicable.
CONSENT FOR PUBLICATION
Written consent has been obtained from the subject.
STANDARDS FOR REPORTING
CARE guidelines were followed.
AVAILABILITY OF DATA AND MATERIALS
The data used in this study are available from the corresponding author [M.A] upon request.
FUNDING
None.
CONFLICT OF INTEREST
The author declares no conflict of interest, financial or otherwise.
ACKNOWLEDGEMENTS
Declared none.


