All published articles of this journal are available on ScienceDirect.
Use of the Breathlessness, Cough, and Sputum Scale (BCSS©) in Pulmonary Rehabilitation§
Abstract
Background:
In pulmonary rehabilitation (PR) effective measures have been taken while in analyzing a patient’s intervention with the help of entry to exit evaluations. The absence of an objective and quantifiable scale are limitations of PR that allow analyzing of a patient’s self reported symptoms throughout PR. The Breathlessness, Cough and Sputum Scale (BCSS©) is used to predict patient exacerbations by evaluating common symptoms identified in the COPD population. This study used the BCSS© survey to track complex symptom changes throughout the course of PR intervention. The BCSS© tool measured the patient’s self reported symptoms in real time for each visit when patient enrolled in PR.
Methods:
Thirty-five patients with COPD from three outpatient PR centers were asked to report the severity of breathlessness, cough, and sputum prior to each PR session using the BCSS© survey.
Results:
There was a significant decrease in self reported symptoms of the mean BCSS© score from entry 4.6(± 2.9) to exit 2.3 (± 2.5), p < 0.001. The results showed variable decrease in the self reported symptoms with more PR visits. The secondary outcome showed high correlations with quality of life measures using the Pulmonary Function Status Scale (PFSS) on entry and exit to PR.
Conclusions:
The BCSS© tool is an effective means for measuring the impact of PR on improving patient tolerance and self-reported symptoms as a result of COPD. More research is needed to better assess the complex symptoms of COPD patients in PR to enhance programmatic outcomes.
INTRODUCTION
According to the American Association of Cardiopulmonary Rehabilitation (AACVPR), Pulmonary Rehabilitation (PR) is a multidisciplinary program to facilitate those with lung disease by helping patients to manage physically, behaviorally, and socially [1, 2]. The American Thoracic Society and European Respiratory Society define PR as:
“An evidence-based, multidisciplinary, and comprehensive intervention for patients with chronic respiratory diseases, who are symptomatic and often have decreased daily life activities. Integrated into the individualized treatment of the patient, pulmonary rehabilitation is designed to reduce symptoms, optimize functional status, increase participation, and reduce health care costs through stabilizing or reversing systemic manifestations of the disease” [2, Page 1].
PR has been established to improve the prognosis of patients with chronic obstructive pulmonary disease (COPD) through educational, psychosocial, and exercise training interventions [2]. Earlier PR research has shown to decrease hospital visits, improve functional capacity, reduce in dyspnea ratings, and increase quality of life measures [2].
Many PR programs implement common outcome assessments for quality improvement in a pre-test/ post-test design such as the Pulmonary Functional Status Scale (PFSS) [3, 4], the COPD Self-Efficacy Scale (COPD-SE) [5, 6], and the six-minute walk test (6MWT) [1]. The COPD-SE scores have significantly improved when patients participate in PR as well as PFSS scores [4, 6]. However, sometimes the PFSS and COPD-SE instruments may not be sensitive enough to detect the symptomatic changes in patients and the scores may not be significantly different from baseline. To address the limitations of psychometric questionnaires, PR programs have recently implemented the B.O.D.E. index based on more physiological parameters using FEV1, 6-Minute Walk Distances, Body Mass Index (BMI), and Modified Medical Research Council (MMRC) dyspnea scale to score a patient’s prognosis [7-9]. Although the above results are important when it comes to assessing the importance of PR intervention, the results only relate entry and exit visits to PR of the COPD patient on entry and exit visits to PR. The B.O.D.E. provides an in-depth scoring system of the patient’s severity of disease and takes into account nutritional status based on BMI [8]. Additionally, the well established pre-test/post-test design applied in many aspects of healthcare programming may not track the subtle symptomatic changes in COPD patients experienced during the course of PR; thus, providing false conclusions that PR is not effective in symptom management.
The benefits of PR may not be fully appreciated with only a pre-test/post-test design. The beginning stages of an exacerbation, especially at the end of PR intervention may result in negative effects, which may in turn prove to be detrimental to PR [9,10]. The Breathlessness, Cough, and Sputum Scale (BCSS©) was developed to provide a quick and easy method of evaluating the severity of respiratory symptoms common in COPD patients [9, 10]. The BCSS© is based on a three-item questionnaire assessing the patient’s breathlessness, cough, and sputum.
The BCSS© allows subjects to record symptoms in a Likert scale having a format with zero representing the improvement symptoms and a 4 indicating worsening of symptoms [10]. The BCSS© easily shows PR staff if the patient’s perception of the symptoms increases from visit to visit. To this date, there has not been a study that evaluates the use of the BCSS© in a PR program. It was the intent of this research study to investigate whether the use of this tool could quantify the subjective, self-reported changes by patients participating in PR.
MATERIALS AND METHODS
Study Population
Thirty-five men and women aged 55-85 years old participating in PR with a primary diagnosis of COPD, were recruited for the study. All patients were referred to PR with qualifying chronic lung disease diagnosis (FEV1 <60% of predicted) as required by institutional policy and insurance reimbursement. Pulmonary function tests which were required to ensure chronic lung disease status, were conducted within 90-days of starting PR. Patients demonstrating Restrictive Lung Diseases, Interstitial Fibrosis, Pneumoconiosis, Rheumatoid Pulmonary Disease, Sarcoidosis, Kyphoscoliosis, Collagen Vascular Lung Disorder, and Poliomyelitis were excluded from the study. Patients who smoke were restricted not to smoke three months prior to the starting date of PR to participate. All participants performed an individualized treatment program (ITP) following specified policies and procedures established by the American College of Chest Physicians (ACCP) and the American Association of Cardiovascular and Pulmonary Rehabilitation (AACVPR) [1, 2]. The emphasis on ITP’s is essential; however, the ITP has been certified by the AACVPR to ensure proper implementation for each PR prescription of care to meet minimum standards of care. Generally, the hospital’s PR program was conducted as follows: a warm-up period with stretching for 5-minutes, exercise training for 45-minutes, a cool-down period for 5-minutes, and educational lectures on disease management for 15-minutes. The study received institutional review board approval. All participants provided written consent to participate in the study.
Outcome Measures
The Breathlessness, Cough, and Sputum Scale, Copyright 2003, was used with permission from the AstraZeneca Company. The BCSS© survey was administered at the beginning of each PR visit. The PFSS quality of life assessment, COPD-SE behavioral assessment, and the 6MWT were completed in the traditional pre-test/post-test design on the first and last day of the patient’s PR program.
Statistical Analysis
Outcomes were analyzed using the Statistical Analysis System (SAS Version 9.0, Cary, NC). Baseline demographic data was assessed for spirometry for disease status (FEV1), 6MWT distance, age, and BMI categories. T-tests were used to monitor differences in BCSS© entry-PR scores and BCSS© exit-PR scores. A mixed model procedure was applied to test the effects of sex (male or female), time (baseline visit to 20-visits over eight weeks), and interactions of these variables with the BCSS© scores. The mixed model procedure was also employed to examine the relationship between changes that occurred in other common outcomes for PR programs using the typical pre-test/post-test design; PFSS quality of life assessment, COPD-SE behavioral assessment, and 6MWT distance functional assessment. A value for P < 0.05 was considered to be statistically significant.
RESULTS
The final data set was composed of 12 (44%) men and 15 (56%) women. Six (24%) subjects were of normal-weight and 19 (76%) were overweight. Significant effects of BMI were found and adjusted P values were used to compare LSMEANS between normal and overweight individuals (adjusted P = 1 – (1 – P)2). Data given in the text, figure and tables are LS Means (± SEM) from the mixed models unless stated otherwise. Baseline characteristics of the subjects are presented in Table 1. There were no differences in baseline 6MWT distance and, PFSS scores, FEV1 values or COPD-SE ratings, between the BMI categories (all P’s > 0.05).
Baseline Subject Characteristics (n = 27)
| Variable | Mean (± SD) | Median (Range) |
|---|---|---|
| Age (y) | 73.1 ± 8.4 | 55 – 84 |
| Weight (kg) | 80.2 ± 17.2 | 52 – 114 |
| Height (cm) | 164.6 ± 7.7 | 152 – 179 |
| Body Mass Index (kg/m2) | 29.5 ± 6.0 | 22 – 43 |
| FEV1:
Forced Expiratory Volume
in the first second, % predicted
FEV1: Forced Expiratory Volume in the first second, L absolute |
7 l ±6.0 0.91 ±0.43 |
30 - 59% 0.43 – 2.23 |
Data are presented as mean ± standard deviation, years (y), kilograms (kg), centimeters (cm), kilogram/meters squared ((kg/m2), percent (%), or liters (L).
Other Typical Outcome Measures in PR Compared to BCSS© (n = 27)
| Variable | Baseline PR Mean (± SD) | Post PR Mean (± SD) | p Value* |
|---|---|---|---|
|
|
|||
| Breathlessness, Cough, and Sputum Scale (BCSS©) | 4.6 ± 2.9 | 2.3 ± 2.5 | 0.007 |
|
|
|||
| Pulmonary Function Status Scale (PFSS) | 80.2 ± 17.2 | 80.2 ± 17.2 | 0.08 |
| COPD Self-Efficacy Scale (COPD-SE) | 73.9 ± 32.0 | 83.4 ± 27.0 | 0.0026 |
| Six-Minute Walk Test (6MWT) (m) | 250.8 ± 120.5 | 300.7 ± 121.2 | < 0.0001 |
Data are presented as mean raw scores from the various instruments, Six-Minute Walk Test distance in meters (m), and ± standard deviation.
* Student’s t-test or general linear model for effects of time.
Breathlessness, Cough, and Sputum Scale (BCSS)©
There was a significant two-way interaction of BMI status and time (P = 0.045) (Fig. 1). Within the Overweight/Obese BMI group, there were significant differences noted between midpoint (visit 10) and endpoint (visit 20). The data reflects a baseline BCSS© score 4.6(± 2.9) to midpoint 3.5 (± 2.2), p=0.009; a midpoint BCSS© score 3.5 (± 2.2) to endpoint 2.3 (± 2.5), p=0.007; and baseline BCSS© score 4.6(± 2.9) to endpoint 2.3 (± 2.5), p < 0.001. No significant differences in symptom ratings were noted between these time points for the Normal BMI group. Furthermore, there were no differences between BMI categories at any time points (all P’s > 0.1). The results of our study have indicated that PR intervention with implementation of the BCSS© provided significant reductions of symptoms thus reducing the risk of exacerbations in most cases. However, anecdotal responses from PR staff indicated a heightened awareness among the patients regarding symptoms when scoring.. The highest total values reported were observed during the exacerbation and warranted calls to physicians. Then, subsequent to physician treatment and medication, the PR staff noticed reductions in scores which is in concert with previously published data [10, 11].
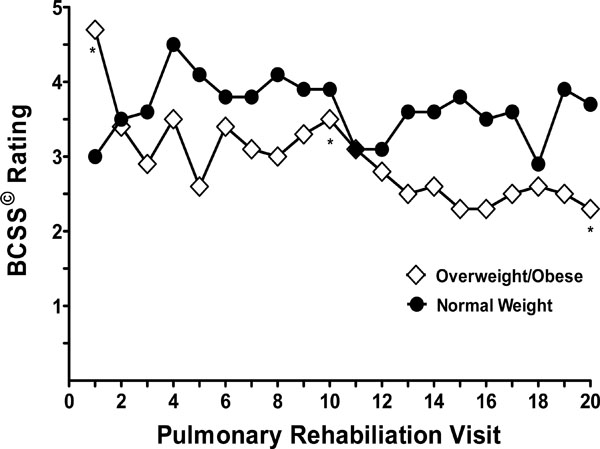
Breathlessness, Cough and Sputum Scale (BCSS) ratings (LS Means) at specified visits to pulmonary rehabilitation by BMI category (Overweight/Obese, BMI > 24.9 kg/m2 versus Normal, BMI < 25 kg/m2). A significant two-way interaction was found between BMI status and time on BCSS ratings (P = 0.045). * Significant differences within the Overweight/Obese group in pairwise comparisons (P’s < 0.01) between baseline, midpoint and endpoint.
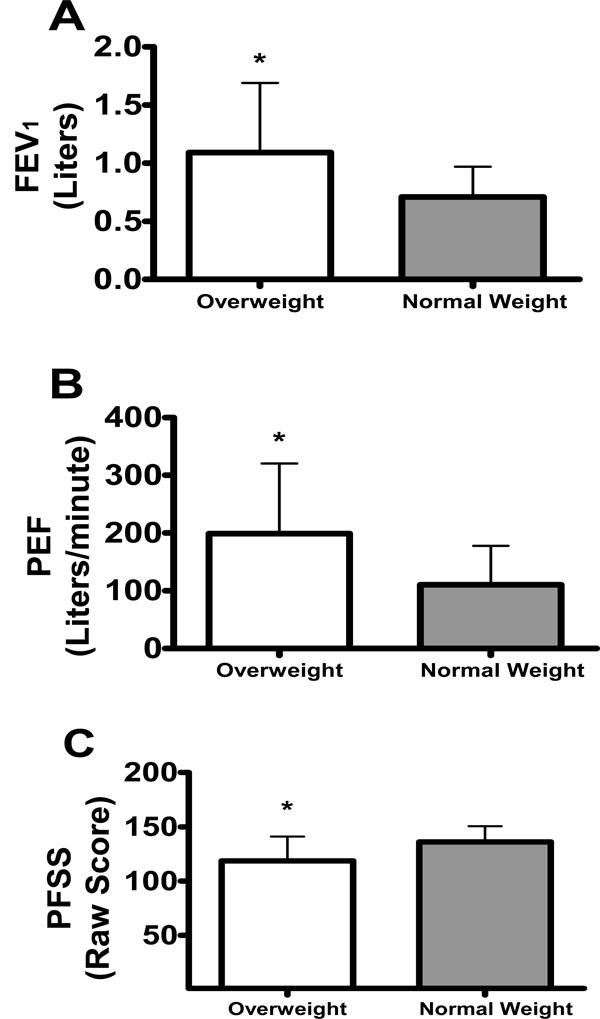
Effect of Body Mass Index (BMI) status on Forced Expiratory Volume in One Second (FEV1) Liters, Peak Expiratory Flow (PEF) Liters/minute, and Pulmonary Function Status Scale (PFSS) total raw score. *Denotes significant values (p<0.05).
When we included subjects with a BMI < 18.5 kg/m2 (n = 3), the two-way interaction of BMI status and time on BCSS© ratings remained significant (p = 0.037). In the higher-BMI group, ratings decreased about 52% between baseline and visit 20, while in the lower-BMI group, this pattern was not apparent (no difference found in scores between baseline and visit 20). Similar to the B.O.D.E. index, BMI status was found to be of significant importance in relation to self-reported symptoms in the BCSS© total scores which indicates that nutritional status must be considered when developing ITP for patients enrolled in PR [7, 12].
Pulmonary Function Status Scale (PFSS)
Although not significant, trends were found for main effects of time (p = 0.08) (Table 2) and BMI category (p = 0.051), where scores decreased by 9% between baseline and end point, and overweight subjects scored approximately 8% lower on average than normal weight subjects (Fig. 2).
Chronic Obstructive Pulmonary Disease – Self-Efficacy Scale (COPD-SE)
There was a main effect of time on COPD-SE where subjects had improved scores at endpoint as compared to baseline (73.9 ± 32.0 versus 83.4 ± 27.0, p = 0.0026) (Table 2).
Six-Minute Walk Test (6MWT)
There was a main effect of time on 6MWT distance where subjects were able to cover less distance at baseline 250.8 ± 120.5 as compared to endpoint 300.7 ± 121.2 meters, p < 0.0001) (Table 2).
Patient Data
During this time, only one patient was reported to have exacerbated during the course of PR. The number of medication changes were n=3 for one time changes to inhaled steroid treatment and beginning of antibiotic treatment.
DISCUSSION
The results of this study displayed that PR intervention with implementation of the BCSS© provided significant reductions of symptoms, thus reducing the risk of exacerbations in most cases. Currently, PR programs are facing strict regulations and criticisms from the Centers for Medicare and Medicaid Services (CMS). Recently, CMS imposed strict regulations contrary to the intent of such outpatient services, especially in the new age of healthcare transformation. With accountable care demonstration projects beginning on January 1, 2012, outcome tools sensitive enough to measure changes in preventive and maintenance care have become essential. The intent of this study was to investigate whether the use of the BCSS© could quantify daily symptom changes of patients participating in PR. According to Leidy et al., there are hardly any instruments through which common respiratory symptoms can be evaluated that may negatively alter the PR outcomes in the COPD population [11]. PR programs do not quantitatively assess three common symptoms to COPD such as breathlessness, cough, and sputum together. This type of research increased the importance of PR programs which are necessary to assist in chronic disease management. According to Leidy, the scale is used to predict worsening of the COPD patient’s condition [10,11]. Preventive services such as pulmonary rehabilitation or physical rehabilitation show a reduction in healthcare costs via reduced re-admissions and exacerbations; however, PR can assess day to day chronic disease management and predict declining health with the BCSS©. The BCSS© has reliably demonstrated significant results correlating changes in disease states of 1,426 pulmonary patients. The analysis of the BCSS© demonstrated that total values obtained were able to predict the improvement of symptoms over the course of PR [11]. The results indicated that even after only 10 visits, self-reported symptoms were significantly reduced as well as continued significant changes from 10 to 20 visits. The BCSS© helps to identify changes that may occur during the treatment of PR that have not been captured before. The information gained by this study provides a glimpse at the daily impact of PR using the BCSS© which may be influential to CMS regulations for predictive and preventive care.
PR programs should implement daily quantifiable, validated, and reliable outcomes in conjunction with other daily measures (e.g. oxygen saturation, dyspnea scale, etc.) and common disease specific tools (e.g. PFSS, the UCSD, the CRQD, and the SGRQ). These outcome measures are commonly utilized at the entry PR evaluation and exit (discharge) PR evaluation. The most commonly used self reported outcome measures, such as quality of life measures, exercise intensity scales, and perceived dyspnea scales help to provide an overall picture of PR intervention [12]. While these tools do provide information regarding the role of PR on quality of life, these tools are not utilized daily and incorporated throughout the PR ITP. This study provides evidence that the BCSS© can track these changes over a course of treatment intervention in PR, which is essential for patient education and management of the disease. The BCSS© scores from this study provide new and novel techniques for measuring clinically significant changes in a PR program to emphasize the clinical necessity of this type of intervention for our COPD population. Rarely, clinicians find such a positive responsive to improvements in a chronic condition such as COPD. This research furnishes along with PR staff another easy-to-use tool for measuring outcomes for quality improvement measures as well as for program certification. As healthcare moves towards Accountable Care Organizations, the information from this study underscores the benefits of PR as a preventive and maintenance service to the COPD population; thus, reducing the burden on an already strained system. Continued efforts should be done to show the benefits and impact of services such as PR using a simple tool yet provides a big picture of PR necessity in today’s accountable health care environment. More research is needed to better assess the complex symptoms of COPD patients in PR to enhance programmatic outcomes.
NOTES
§ Study conducted in the Cleveland Clinic Health System in Cleveland, Ohio. This manuscript has not been published and is not under consideration elsewhere in whole or any language except as an abstract.
CONFLICT OF INTEREST
The authors confirm that this article content has no conflict of interest.
ACKNOWLEDGEMENTS
Declared none.


