All published articles of this journal are available on ScienceDirect.
Repeatability of the Evaluation of Perception of Dyspnea in Normal Subjects Assessed Through Inspiratory Resistive Loads
Abstract
Purpose:
Study the repeatability of the evaluation of the perception of dyspnea using an inspiratory resistive loading system in healthy subjects.
Methods:
We designed a cross sectional study conducted in individuals aged 18 years and older. Perception of dyspnea was assessed using an inspiratory resistive load system. Dyspnea was assessed during ventilation at rest and at increasing resistive loads (0.6, 6.7, 15, 25, 46.7, 67, 78 and returning to 0.6 cm H2O/L/s). After breathing in at each level of resistive load for two minutes, the subject rated the dyspnea using the Borg scale. Subjects were tested twice (intervals from 2 to 7 days).
Results:
Testing included 16 Caucasian individuals (8 male and 8 female, mean age: 36 years). The median scores for dyspnea rating in the first test were 0 at resting ventilation and 0, 2, 3, 4, 5, 7, 7 and 1 point, respectively, with increasing loads. The median scores in the second test were 0 at resting and 0, 0, 2, 2, 3, 4, 4 and 0.5 points, respectively. The intra-class correlation coefficient was 0.57, 0.80, 0.74, 0.80, 0.83, 0.86, 0.91, and 0.92 for each resistive load, respectively. In a generalized linear model analysis, there was a statistically significant difference between the levels of resistive loads (p<0.001) and between tests (p=0.003). Dyspnea scores were significantly lower in the second test.
Conclusion:
The agreement between the two tests of the perception of dyspnea was only moderate and dyspnea scores were lower in the second test. These findings suggest a learning effect or an effect that could be at least partly attributed to desensitization of dyspnea sensation in the brain.
INTRODUCTION
Dyspnea is a subjective experience of breathing discomfort that consists of qualitatively distinct sensations that vary in intensity [1-3]. The perception of dyspnea does not correlate with clinical severity or with the underlying pathology and may be affected by emotional, behavioral and cultural influences and environmental factors [4]. The investigation of the perceived level of dyspnea experienced by patients aims to make it possible to detect those individuals with abnormal sensitivity to breathlessness [5]. Asthma patients with low perception of dyspnea have higher morbidity and mortality due to delay in seeking medical care [5-7]. Identifying these patients would help to establish early treatment, improving survival and reducing health costs [8].
Several studies have used a system based on inspiratory resistive loads to evaluate the perception of dyspnea and to investigate factors associated with increased or decreased sensitivity to breathlessness [5, 7, 9-11]. This system for evaluating the perception of dyspnea consists of a circuit with inspiratory resistive loads of increasing magnitudes that induces the sensation of dyspnea by increasing the effort and work of breathing, while the individual expresses the degree of shortness of breath using a scale, such as the Borg scale [12, 13]. The breathing pattern during the test can influence the magnitude of the perception of dyspnea [14], just as the increase of the inspiratory resistive loads can influence the breathing pattern. The dyspnea induced by the devices used in the laboratory can differ between individuals, even in healthy subjects [9,15]. Furthermore, hypoxia can suppress the sensation of dyspnea in asthmatics [10].
Repeatability and reproducibility of a method are essential factors to ensure reliable results for diagnostic use in clinical practice. Repeatability (r) refers to the strength of agreement between repeated measurements obtained under similar operating circumstances, with the same operator, and with the same equipment. It can be expressed by the following equation: where σ1 corresponds to the standard deviation. Reproducibility (R) is the strength of agreement between repeated measurements obtained with the application of the same method under different operating conditions, by different operators, or with different equipment. It can be expressed by the following equation: where σ2 represents the standard deviation related to all other causes of variability of results not related to the repeatability [10,16].
Although there are several reports investigating factors related to the variability of the perception of dyspnea assessed by inspiratory resistive load system, there is insufficient and conflicting evidence regarding the repeatability of the method [17,18].
The objective of this study was to investigate the repeatability of the method to evaluate the perception of dyspnea in normal subjects during ventilation in an inspiratory resistive load system.
METHODS
Study Design
We conducted a cross-sectional study with data collected prospectively to evaluate the repeatability of the perception of dyspnea in healthy subjects. The volunteers underwent two tests with an interval of time from 2 to 7 days between tests.
The study was approved by the Hospital de Clínicas de Porto Alegre (HCPA) ethics committee and was registered with the number 08-064. All individuals studied had previously signed the Informed Consent Form.
Population
The study was conducted in the Service of Pneumology, HCPA, Porto Alegre, Brazil and included healthy subjects ≥18 years of age. The volunteers were recruited with notices posted in the hospital and electronic announcements. The females that were of childbearing potential were allowed to be enrolled in the study as long as they were practicing a highly effective method of contraception (oral, injectable or implanted hormonal methods of contraception, placement of an intrauterine device or intrauterine system condom or occlusive cap with spermicidal foam/gel/film/cream/ suppository, male sterilization, or true abstinence). Acute respiratory tract complaints in the last 30 days, current or past smoking, abnormal spirometry or any chronic medical conditions such as asthma, chronic pain, cardiac or orthopedic disease were exclusion criteria. In addition, volunteers were excluded if they had traumatic injury.
Study Procedures
All subjects included in the study were submitted to a routine clinical evaluation conducted by the medical members of the research team. A standardized form was used for evaluating and recording the data. Pulmonary function tests were measured with a Pony graphic 4.0 spirometer (Cosmed Srl, Pavona di Albano, Roma, Italy). The forced vital capacity (FVC), forced expiratory volume in one second (FEV1) and FEV1/FVC were measured three times, and the best trial was recorded. All results were expressed in liters and as percent predicted for age, height and sex [19,20].
The perception of dyspnea was evaluated through an inspiratory resistive loads system, using a model previously reported in the literature [5] comprised of a two-way non-rebreathing valve (Hans-Rudolph, Kansas, USA). A plastic circular mouthpiece, with eight different orifices generated the inspiratory loads of increasing magnitude (6.7, 15.0, 25.0, 46.7, 67.0 and 78.0 cm H2O/L per second, calculated according to a constant flow of 300 mL/s) (Fig. 1). The intrinsic resistance of the system was 0.6 cm H2O/L per second. Before the test, participants were familiarized with the apparatus and measurement procedures. After standardized instructions, volunteers were seated in a comfortable chair. Wearing a nose clip, volunteers breathed through a mouthpiece in the resistive loads system. The sensation of dyspnea was assessed during ventilation with increasing inspiratory resistive loads. After breathing at rest and at each level of resistance for two minutes, the subjects were questioned about the feeling of shortness of breath (dyspnea) experienced during the test using the modified Borg scale [13], ranging from 0 (no dyspnea) to 10 (maximal dyspnea). The heart rate and the peripheral oxygen saturation (SpO2) were measured by pulse oximetry (NPB-40; Nellcor Puritan Bennett; Pleasanton; USA) during the test. To monitor the effects of dyspnea stimulation, inspiratory pressure, inspiratory time and respiratory frequency were measured continuously at the mouthpiece using computer software. Expiratory load was not applied. Subjects were free to choose their breathing rate, volume and flow to have as natural a breathing pattern as possible. Volunteers repeated the test with the same operator, same equipment and same method in a time interval from 2 to 7 days between tests.
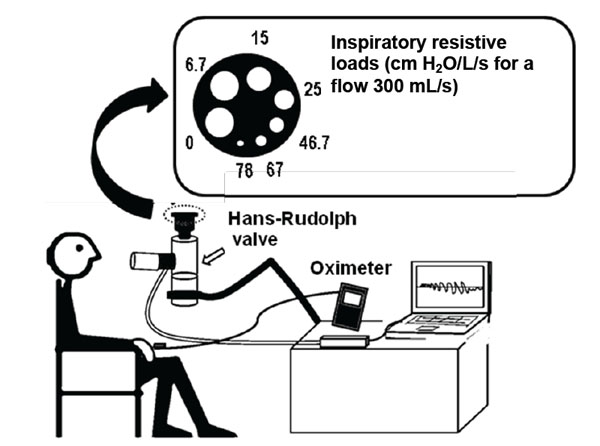
Inspiratory resistive load system.

Generalized Linear Model Between Two Tests for Perception of Dyspnea Scores. There was a significant difference between the steps of resistive loads (p <0.001) and between the tests (p =0.003).
Correlations Between Borg Dyspnea Scores and Inspiratory Pressures for Both Resistive Loads Tests (r=0.49 and p <0.001, for the first test; r=0.36 and p <0.001).

Kaplan-Meier Analysis of Proportion of Subjects that Completed Resistive Loads Testing (log rank test=0.647).
Statistical Analysis
Data analysis was carried out with the Statistical Package for the Social Sciences (SPSS) version 18.0 (Chicago, Illinois) and Medcalc program, version 10.2.0 (MedCalc Software, Mariakerke, Belgium). Data were expressed as number and proportion of cases, mean ± standard deviation (SD) or median (interquartile range).
Measurement variation was quantified as the within-subject coefficient of variation, the intraclass correlation coefficient for average measures, or the weighted kappa coefficient of agreement (linear set of weight), as appropriate. The root mean square method was used to calculate the coefficient of variation, as proposed by Bland. The coefficient of variation was found separately for each subject, then squared, the mean determined, and the square root of that mean was taken. Confidence intervals were calculated for coefficient of variation and intra-class correlation coefficient.
Generalized linear models were used to compare the dyspnea scores, inspiratory pressures and respiratory frequencies between the two tests during the inspiratory resistive loads. Correlations were determined using Spearman’s rank correlation coefficient. Kaplan-Meier curves, stratified by test, were compared with the log rank test, to compare the completion of the perception of dyspnea test during inspiratory resistive loads.
The statistical significance level was set at p<0.05. All probabilities reported were two-tailed.
RESULTS
The study included 16 Caucasian individuals, 8 men and 8 women. The mean age was 36.3 ± 11.9 years (19-61 years) and the mean body mass index (BMI) was 23.9 ± 2.8 kg/m2 (18.7 to 28.7 kg/m2). Nine subjects practiced physical activity ≥ 2 times/week. Most individuals had completed higher educational level. The mean FEV1 was 98.75 ± 9.75% of predicted and the mean FVC was 95.31 ± 10.12% of predicted (Table 1).
Demographic characteristics and pulmonary function of subjects.
| Characteristics | |
|---|---|
| Subjects, n | 16 |
| Age (years), mean ± SD | 36.3 ± 11.9 |
| Sex (male/female), n | 8/8 |
| Body mass index (kg/m2), mean ± SD | 23.9 ± 2.8 |
| Race (Caucasian), n | 16 |
| Educational level, n (%) | |
| ≤ 8 years of school | 2 (12.5%) |
| > 9 years school and < high school | 2 (12.5%) |
| ≥ high school | 12 (75.1) |
| Physical activity ≥ 2x/week | 9 (56.3%) |
| FEV1 (L), mean ± SD | 3.5 ± 0.7 |
| FEV1 (% predict)*, mean ± SD | 98.8 ± 9.8 |
| FVC (L), mean ± SD | 3.8 ± 0.7 |
| FVC (% predict)*, mean ± SD | 95.3 ± 10.1 |
n = number of cases; SD = Standard deviation; PEFR = peak expiratory flow rate; FEV1 = forced expiratory volume in one second, FVC = forced vital capacity.
*Percent predicted for age, height and sex (Pereira, et al.: J Bras Pneumol 2007 Aug; 33(4): 397- 406).
Measurement variation of dyspnea scores between the two resistive loads tests.
| Loads (cm H2O/L/S) | 1° Test | 2° Test | SE | Kw | Kw 95%CI | ICC | ICC 95%CI | ||
|---|---|---|---|---|---|---|---|---|---|
| Mean ± SD | Median (IR) | Mean± SD | Median (IR) | ||||||
| @ 0 | 0.9 ± 1.3 | 0 (2.0) | 1 ± 1 | 0 (2.0) | 0.080 | 0.78 | 0.62- 0.94 | 0.88 | 0.70-0.96 |
| 6.7 | 1.9 ± 1.7 | 2 (2.5) | 2 ± 2 | 0 (3.0) | 0.117 | 0.46 | 0.23 - 0.69 | 0.69 | 0.31-0.88 |
| 15 | 2.9 ± 2.5 | 3 (3.3) | 2 ± 3 | 2 (3.0) | 0.105 | 0.58 | 0.38 - 0.79 | 0.90 | 0.73-0.96 |
| 25 | 4 ± 3.0 | 4 (3.0) | 2 ± 2 | 2 (3.0) | 0.112 | 0.33 | 0.11 - 0.55 | 0.63 | 0.19-0.87 |
| 46.7 | 4 ± 2.0 | 5 (5.0) | 3 ± 2 | 3 (4.0) | 0.089 | 0.57 | 0.39 - 0.74 | 0.93 | 0.79-0.98 |
| 67 | 5 ± 3.0 | 7 (5.0) | 4 ± 3 | 4 (5.0) | 0.096 | 0.56 | 0.37 - 0.74 | 0.80 | 0.48-0.93 |
| 78 | 5 ± 3.0 | 7 (5.0) | 4 ± 3 | 4 (5.0) | 0.078 | 0.73 | 0.58 - 0.89 | 0.95 | 0.83-0.98 |
| @ 0 | 2 ± 2.0 | 1 (1.0) | 1.1 ± 1.6 | 0.5 (2.0) | 0.162 | 0.52 | 0.20 - 0.84 | 0.74 | 0.34-0.91 |
SD = standard deviation; IR= interquartile range; SE= standard error; CI = 95% confidence interval; Kw = weighted Kappa coefficient of agreement; ICC = intraclass correlation coefficient for average measures.
Measurement variation of inspiratory pressures and of respiratory frequency between the two resistive loads tests.
a. Inspiratory Pressures.
| Loads (cm H2O/L/S) | 1° Test | 2° Test | CV (%) | CV 95%CI (%) | ICC | ICC 95%CI |
|---|---|---|---|---|---|---|
| Mean ± SD | Mean ± SD | |||||
| @ 0 | 3.2 ± 1.8 | 3.9 ± 1.9 | 38.8 | 5.7 - 54.6 | 0.57 | 0.126-0.827 |
| 6.7 | 5.4 ± 3.4 | 5.4 ± 2.8 | 28.5 | 16.6 - 36.8 | 0.80 | 0.525-0.927 |
| 15 | 8.8 ± 5.6 | 7.6 ± 4.3 | 28.8 | 20.1 - 35.5 | 0.74 | 0.407-0.902 |
| 25 | 12.5 ± 8.4 | 10.2 ± 5.7 | 25.8 | 17.2 - 32.1 | 0.80 | 0.499-0.927 |
| 46.7 | 14.4 ± 10.9 | 12.3 ± 7.9 | 21.8 | 12.9 - 27.9 | 0.83 | 0.541-0.940 |
| 67 | 15.7 ± 12.3 | 14.7 ± 0.6 | 22.7 | 11.9 - 29.8 | 0.86 | 0.609-0.951 |
| 78 | 14.6 ± 9.9 | 16.3 ± 2.2 | 21.9 | 15.5 - 26.7 | 0.91 | 0.735-0.972 |
| @ 0 | 4.4 ± 2.8 | 4.7 ± 2.8 | 20.2 | 9.6 - 26.9 | 0.92 | 0.751-0.974 |
b. Respiratory Frequency (Breaths Over 2 min).
| Loads (cm H2O/L/S) | 1° Test | 2° Test | CV (%) | CV 95%CI (%) | ICC | ICC 95%CI |
|---|---|---|---|---|---|---|
| Mean ± SD | Mean ± SD | |||||
| @ 0 | 25.1 ± 11.9 | 29.1 ± 20.4 | 19.9 | 12.8 - 25.1 | 0.74 | 0.399-0.900 |
| 6.7 | 23.3 ± 17.8 | 25.8 ± 17.1 | 22.2 | 10.0 - 29.7 | 0.94 | 0.844-0.980 |
| 15 | 24.7 ± 18.7 | 24.1 ± 14.0 | 14.9 | 8.2 - 19.4 | 0.92 | 0.791-0.972 |
| 25 | 24.1 ± 18.7 | 20.9 ± 7.8 | 22.6 | 6.8 - 31.3 | 0.60 | 0.149-0.846 |
| 46.7 | 20.7 ± 8.7 | 19.8 ± 8.7 | 31.8 | 8.2 - 44.2 | 0.57 | 0.075-0.837 |
| 67 | 22.4 ± 13.9 | 18.3 ± 6.7 | 22.9 | 10.6 - 30.7 | 0.62 | 0.155-0.860 |
| 78 | 21.2 ± 10.2 | 20.1 ± 7.9 | 18.3 | 11.0 - 141.5 | 0.70 | 0.272-0.899 |
| @ 0 | 21.5 ± 10.3 | 20.9 ± 9.7 | 12.5 | 7.7 - 140.7 | 0.95 | 0.855-0.986 |
SD = standard deviation; CV = coefficient of variation; CI = 95% confidence interval; ICC = intraclass correlation coefficient for average measures.
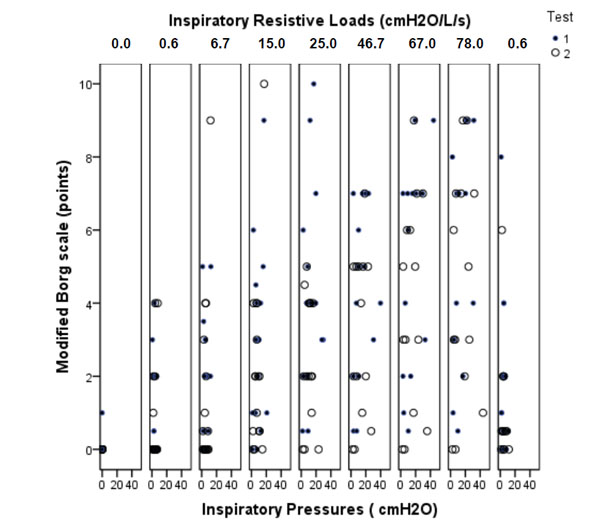
Table 2 presents the variation in the measurement of dyspnea scores between the two resistive loads tests. The median scores in the first test were 0, 2, 3, 4, 5, 7, 7 and 1 for the steps of application of inspiratory loads of ( 0; 6.7; 15.0; 25.0; 46.7; 67.0, 78.0 and ≅ 0 cm H2O/L per second, respectively. The median scores in the second test were 0, 0, 2, 2, 3, 4, 4 and 0.5 points, respectively. The kappa coefficient of agreement was 0.78, 0.46, 0.58, 0.33, 0.57, 0.56, 0.73 and 0.52 for each point, respectively. The intra-class correlation coefficient was 0.88, 0.69, 0.90, 0.63, 0.93, 0.80, 0.95 and 0.74 for each load, respectively.
Fig. (2) shows generalized linear models for Borg dyspnea scores during increasing steps of resistive loads between the two tests. There was a statistically significant difference between the steps of resistive loads (p <0.001) and between the tests (p =0.003). Values of dyspnea scores were significantly lower in the second test.
In the first test, the mean inspiratory pressures generated in each step of inspiratory loads were 3.2, 5.4, 8.8, 12.5, 14.4, 15.7, 14.6 and 4.4 cmH2O, respectively. In the second test, the mean inspiratory pressures were 3.9, 5.4, 7.6, 10.2, 12.3, 14.7, 16.3 and 4.7 cmH2O, respectively. The intra-class correlation coefficients were 0.57, 0.80, 0.74, 0.80, 0.83, 0.86, 0.91and 0.92 for each step, respectively (Table 3). In the analysis by the generalized linear model, as the magnitude of the loads increased, mean inspiratory pressure increased significantly (p<0.001), but without a difference between groups (p=0.59).
In the first test, the mean respiratory frequencies (breaths over 2 min) observed in each step of inspiratory loads were 25.1, 23.3, 24.7, 24.1, 20.7, 22.4, 21.2 and 21.5, respectively. In the second test, the mean respiratory frequencies were: 29.1, 25.8, 24.1, 20.9, 19.8, 18.3, 20.1 and 20.9 breaths over 2 minutes. The intra-class correlation coefficients were for each step: 0.74, 0.94, 0.92, 0.60, 0.57, 0.62, 0.70 and 0.95 (Table 3). In the analysis by generalized linear model, as the magnitude of the loads increased, mean respiratory frequencies decreased significantly (p<0.001), but without significant difference between the tests (p = 0.81).
As the magnitude of the loads increased, there were no significant differences for SpO2 and heart between loads and between tests.
Fig. (3) presents correlations between Borg dyspnea scores and inspiratory pressures during each inspiratory resistive load for both tests. Borg dyspnea scores were significantly correlated with inspiratory pressure (r= 0.49 and p<0.001, for the first test; r=0.36 and p <0.001, for the second test).
Fig. (4) presents the Kaplan-Meier analysis of proportion of subjects that completed resistive loads testing. Thirteen (81.3%) subjects performed the whole evaluation in the first test compared to 14 (87.5%) in the second test (log rank test=0.647). Three individuals drop out of the study in the first test (subject number 5 at inspiratory load of 67 cm H2O/L per second, reporting Borg score of 9; subject number 7 at inspiratory load of 25 cm H2O/L per second, reporting Borg score of 9; subject number 14 at inspiratory load of 25 cm H2O/L per second, reporting Borg score of 10). Two individuals dropped out of the study in the second test (subject number 7 at inspiratory load of 25 cm H2O/L per second, reporting Borg score of 4; and subject number 14 at inspiratory load of 15 cm H2O/L per second, reporting Borg score of 10. The main reasons for dropping out of the study were dyspnea and exhaustion.
DISCUSSION
This study evaluated the repeatability of the evaluation of the perception of dyspnea induced by inspiratory resistive loads in healthy subjects. Repeatability and reproducibility are different measurement conditions which will give rise to different estimates of precision. Repeatability conditions are when replicate measurements are made in one laboratory, by a single analyst, using the same equipment over a short time period. A common definition of reproducibility conditions is when the replicate measurements are made by different analysts, working in different laboratories, using different equipment over an extended time period.
There was moderate agreement in dyspnea scores for each level of resistive loads. Dyspnea scores were significantly lower in the second test. However, the inspiratory pressure generated against various resistive loads and respiratory frequencies did not differ significantly between the tests. These findings could suggest a learning effect through the ventilation system of resistive loads associated with a better control of the sensation of dyspnea by cortical afferents or because the individual became more familiar with the procedure and thereby more comfortable while doing the second test. Otherwise, this finding could be at least partly attributed to desensitization of dyspnea sensation in the brain. Desensitization to dyspnea is often discussed as a mechanism to explain benefit in the rehabilitation of COPD patients. A common hypothesis used to explain the mechanism is alleviation of fear by repetitive performance of exercise tasks in a safe and supervised environment [21-24].
Several studies have shown that dyspnea is multidimensional and that there may be differences between sensory and emotional aspects of its perception [9, 25]. The level of the perception of dyspnea assessed by induction systems of dyspnea differs between people; however, there are few studies assessing the repeatability of these tests [17, 18].
Some studies suggest that dyspnea may be evaluated through the creation of multidimensional scales to assess qualitative aspects of sensory and emotional symptoms [11]. In our study, the method used to measure the perception of dyspnea was the modified Borg scale [13]. The modified Borg scale comprises a simple and validated instrument that has been widely used in clinical practice to evaluate dyspnea [12, 26].
The respiratory rate decreased significantly during breathing through inspiratory resistive loads of increasing magnitude in both tests in relation to the resting situation, which is a typical finding in studies that use resistive loads system, producing increasing respiratory effort [9, 11, 27, 28]. Previous studies demonstrated that changes in ventilatory parameters during exercise are associated with the level of dyspnea [29, 30].
In our study, the perception of dyspnea increased progressively according to the increase of the inspiratory pressure generated at each step of resistive loads, as demonstrated in previous works [11, 14]. However, the novel finding in this study was the recognition that the correlation between dyspnea scores and inspiratory pressures was higher in the first test (r = 0.490) compared to the second one(r = 0.363), which can be attributed to a learning effect and a reduced perception of this symptom.
In the present study, after breathing at each level of resistive load for two minutes without resting, volunteers were asked to rate their feeling of dyspnea. The fact that the test was performed without pausing may explain why the test was discontinued in three patients in the first occasion and in two patients in the second occasion. However, there was no difference in interruptions between the two tests.
Dyspnea perception has been already analyzed mainly during exercise. Stark etal. [31] induced breathlessness by submaximal graded exercise in healthy subjects while objective measurements of cardiorespiratory function were made. Breathlessness was assessed with serial visual analogue scales, but with various measures to enhance repeatability. A high level of reproducibility was obtained in spite of the subjective nature of the assessment. Mador etal. [32] evaluated the 5 weeks reproducibility of Borg scale ratings of the effort to breathe (Borg) and the degree of discomfort evoked by breathing in patients with COPD during exercise. They conclude that during incremental exercise Borg ratings of dyspnea are not as reproducible as physiologic indices in patients with COPD. Grant etal. [33] compared the reproducibility and the sensitivity to change of visual analogue scales, Borg scales, and Likert scales in normal subjects during submaximal exercise. This study suggested that subjective scales can reproducibly measure symptoms during steady-state exercise and can detect the effect of a drug intervention. Also, Chetta etal. [34] studied in asthmatics the score of bronchoconstriction-associated breathlessness at 20% fall in FEV1 evaluated on a Borg scale. They showed a good reproducibility and consider that this allows the serial evaluation of patient's breathlessness perception by this technique in clinical settings and in the physiology laboratory.
The clinical implication of the study is related to the repeated use of the test to assess the same subject over time. In this circumstance, the possibility of the individual becoming acclimated should be considered and may in itself justify a change in the perception of dyspnea. Additional studies are needed to quantify the magnitude and extent of this variation along a greater number of repeated measurements.
The current study has several limitations. The first limitation was the small number of subjects studied. Although the difference between the two tests was statistically significant, the small sample size may have contributed to the large confidence interval for the variation. The second limitation was the fact that only the repeatability (keeping the same conditions, the same operator and a short interval between tests) was studied. The variation of the test could be even higher under reproducibility conditions (different conditions, different operators and longer time interval between the tests). The third limitation was that the subjects studied were exclusively healthy volunteers and the results cannot be extrapolated to disease states. The fourth limitation was that we did not apply a randomized sequence of inspiratory resistive loads. Randomized presentations of different loads might be an alternative method that avoids the perception by patients of the progressive magnitude of the loads.
The agreement between the two tests of the perception of dyspnea induced by inspiratory resistive loads was only moderate and dyspnea scores were lower in the second test. These findings provide evidence for a learning effect or could be at least partly attributed to desensitization of dyspnea sensation in the brain during the repetitive performance of the test. The perception of dyspnea may be modified by previous experience. The subject may develop better control of the sense of cortical afference and/or learn to ventilate in the system with repeated measures. This effect should be considered when repeatedly performing the test over time.
CONFLICT OF INTEREST
The authors confirm that this article content has no conflict of interest.
ACKNOWLEDGEMENTS
We thank Vânia Naomi Hirakata, Statistics Department, Hospital de Clínicas de Porto Alegre, for their support in the statistical analyses.


