All published articles of this journal are available on ScienceDirect.
Outcomes of a Newly Established Electromagnetic Navigation Bronchoscopy Service in the Middle East
Abstract
Aims:
Electromagnetic Navigation Bronchoscopy (EMNB) is a useful tool for the bronchoscopist to target peripheral pulmonary lesions. It has a well-established efficacy and safety profile; however, there are no studies describing its utility in a Middle Eastern population. In this paper, we describe the efficacy and safety outcomes of a newly established EMNB service.
Background:
The diagnosis of peripheral pulmonary lesions presents a significant challenge to the bronchoscopist, especially in the era of increased thoracic imaging with computerized tomography (CT) scans. EMNB is a relatively novel technique that utilizes an image-guided localization system akin to Global Positioning Satellite (GPS) technology, offering the bronchoscopist an accurate navigational pathway to sample peripheral pulmonary targets.
Objective:
We present our initial experience of performing EMNB and report our diagnostic and safety outcomes with EMN bronchoscopy.
Methods:
We conducted a retrospective review of the medical notes of all patients booked for EMNB from May 2015 to December 2019 at our tertiary care center using the electronic medical record system.
Results and Discussion:
Fifty-five patients were scheduled for EMNB, and 47 patients (24 males, 23 females) had EMNB-guided sampling between May 2015 and December 2019. The median age of the patients was 61 years (IQR 49.5-74.3). A bronchus sign was present on the CT chest in 29 (61.7%) cases. Thirty-one (66%) patients had positive EMNB guided samples. There was a weak correlation between the lesion size and the positive EMNB guided sampling (r: 0.34). Twenty-one of 29 (72%) patients with positive bronchus signs had positive EMNB guided samples, compared to 10/18 (56%) patients without bronchus signs; however, the difference was not statistically significant (p-value 0.335). When the presence or absence of rapid onsite examination of cytopathological specimens (ROSE) was compared during the procedure, a trend favoring the presence of ROSE could be seen, but this was statistically non-significant (p-value 0.078). In this series, one patient with pre-existing triple vessel coronary artery disease developed an inferior wall ST-segment elevation myocardial infarction (STEMI), likely secondary to spasm. This patient recovered completely and was discharged from the hospital.
Conclusion:
This study demonstrates that EMNB can be safely performed in a Middle Eastern population with results similar to those reported in major international studies. The highest diagnostic yield was in patients with a bronchus sign on a CT scan, and combining EMNB with ROSE can increase the chances of having a positive diagnostic bronchoscopy. However, patients and physicians need to be aware of the need to follow up with the patients with negative biopsies to ensure that false negatives are not missed.
1. INTRODUCTION
The diagnosis of peripheral pulmonary lesions presents a significant challenge to the bronchoscopist, especially in the era of increased thoracic imaging with chest CT scans. Conventional modalities for the diagnosis of such lesions, such as bronchoscopy with transbronchial biopsy, CT-guided biopsy, and video-assisted thoracoscopic surgery (VATS), are well-established and widely used. However, the inability to accurately guide sampling instruments to the target lesion, concerns for a higher pneumothorax rate, and the invasive nature are some of the limitations of these modalities, highlighting the need for a minimally invasive modality to safely access and sample peripheral pulmonary nodules. Electromagnetic navigation bronchoscopy (EMNB) is a relatively novel technique that utilizes an image-guided localization system akin to a global positioning satellite (GPS) technology, offering the bronchoscopist an accurate navigational pathway to reach peripheral pulmonary targets [1].
One of the two main platforms for performing electromagnetic navigation bronchoscopy is the superdimension navigation system (Covidien, USA). The superdimension device that is used at the authors’ center integrates three separate technologies, enabling the bronchoscopist to navigate the peripheral pulmonary targets accurately. A planning software converts digital imaging from a pre-operative Computerized Tomography (CT) scan into a virtual three-dimensional reconstruction of the tracheobronchial tree, thus enabling a navigational pathway to be created to reach difficult to access regions. An electromagnetic (EM) board, which is a field generator connected to the EMNB console tower containing the planning data, depicts the exact position of a locatable guide (LG) when placed within the EM field on the ENB console monitor. The LG contains a position sensor attached to a steerable probe that has 360-degree maneuverability, enabling guided navigation of the bronchial tree. Once the target is reached, the LG is withdrawn, leaving a Working Channel (WC) guide sheath locked in position. Tools and other catheters can be inserted through the WC for sampling the target lesion [2, 3].
The authors’ hospital is a multispecialty tertiary care hospital in the Middle East that opened its door to patients in 2015. They provide a comprehensive bronchoscopy and interventional pulmonary service ranging from advanced diagnostics to interventional therapeutic procedures for airway and pleural diseases. To our knowledge, this is the only center in our region providing an EMNB service for the diagnosis of peripheral pulmonary lesions. We present our initial experience of performing EMNB in this study.
2. MATERIALS AND METHODS
We conducted a retrospective review of the medical notes of all patient's booked for EMNB from May 2015 to December 2019 at the tertiary care center using the electronic medical record system -EPIC. Ethics approval was obtained from the Cleveland Clinic Abu Dhabi Research Ethics Committee (REC), (REC Number: A-2019-061).
The clinical variables recorded for the study included patient demographics and suspected initial diagnosis. All radiological images were reviewed by a single thoracic radiologist for the purpose of this study, and the characteristics recorded included lesion size, lesion morphology, distance from the origin of the first segmental airway to lesion measured on CT in a straight line, and the distance from the periphery of the lung. The final cytopathological results were recorded. Cytopathological analysis was performed at the pathology laboratory except for molecular markers for lung cancer which were sent for analysis to an outside hospital. Bronchoscopic modalities used during the procedure were recorded, including sampling techniques, the use of radial endobronchial ultrasound (EBUS), fluoroscopy, and rapid onsite examination of cytopathological specimens (ROSE).
The primary outcome of interest was the diagnostic yield of the EMNB-guided bronchoscopic sampling techniques. The secondary outcome was whether EMNB contributed meaningfully to the final diagnosis. This was determined after the case had been discussed in a multidisciplinary meeting involving pulmonologists, oncologists, pathologists, thoracic surgeons, and thoracic radiologists. All adverse events were recorded.
Statistical analysis was performed using Microsoft Excel 2019 for Windows software (with Real Statistics resource pack add-in). Data are expressed as mean ± SD when normally distributed and as the median and interquartile range (IQR) when they are non-normally distributed. Normality of distribution was assessed using the Shapiro–Wilk test. Proportions were used as descriptive statistics for categorical variables. The correlation between parameters was calculated and expressed using Pearson’s coefficient. Comparisons of values between groups of patients were performed by the 1-tailed Student t-test, and p <0.05 was considered statistically significant.
3. RESULTS
Fifty-five patients were scheduled for EMNB, and 47 patients (24 males, 23 females) had EMNB-guided sampling between May 2015 and December 2019. The median age of the patients was 61 years (IQR 49.5-74.3). The median diameter of the lesion on the CT chest was 21.9 mm (IQR 16.5-32.4). Twenty-one of 47 (44%) patients had a spiculated lesion on lung imaging. The lesions were solid in 33 (70.2%) patients, semi-solid in 12 (25.5%), and ground-glass opacities (GGOs) in 2 (4.3%) cases. A bronchus sign was present on the CT chest in 29 (61.7%) cases.
In 40 of 47 (85.1%) patients, a single target was sampled. The most common location of EMNB sampling was the right lower lobe (RLL) (31.9%), followed by the left upper lobe (LUL) (21.3%), the left lower lobe (LLL) (17%), and finally, the right upper lobe (RUL) (14.9%). Fluoroscopy was used in 45 (95%) cases, while radial EBUS was used in 6 (12.8%). During the procedures of 24 patients (51.1%), ROSE was available.
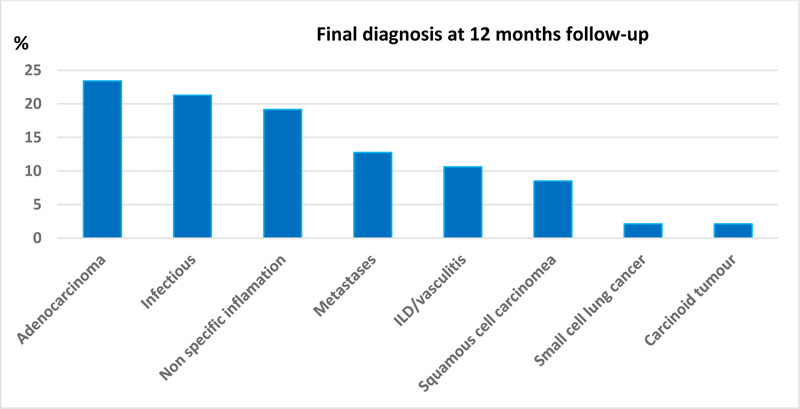
Thirty-one (66%) patients had positive EMNB-guided samples, which included 26 transbronchial biopsies, 10 brushings, and four TBNAs. EMNB contributed meaningfully to the diagnosis in 41 patients (87.2%). Forty-two (89.4%) patients also had non-EMB-guided samples taken during the same procedure, and the same diagnosis was also obtained from non-EMN samples in 12 (25.5%) patients. Only one patient had a positive non-EMN sample when EMN sampling was negative (this patient presented multiple pulmonary nodules and thoracic adenopathy; EBUS-TBNA of mediastinal lymph nodes showed non-caseating granulomas, whereas EMN guided sampling of a pulmonary nodule was non-contributory). Six patients (12.7%) required further invasive procedures (five CT-guided biopsies, one VATS) to achieve a diagnosis. At 12 months post-procedure, the most common final diagnosis was adenocarcinoma (n=11, 23%). Table 1 summarizes the baseline characteristics and results, whereas Fig. (1) demonstrates the final diagnosis at 12 months follow-up.
There was a weak correlation between the lesion size and the positive EMNB-guided sampling (r: 0.34). No correlation was seen between a positive EMN guided sample and age, the distance of the lesion from the origin of the first segmental airway, or the distance of the lesion from the pleura/diaphragm (r: 0.23. -0.13 and 0.11, respectively).
| Characteristics | n | % |
|---|---|---|
| Total | 47 | |
| Male | 24 | 51.1 |
| Female | 23 | 48.9 |
| Age in years - median (IQR) | 61 (49.5-74.3) | |
| Type of Lesion | ||
| Diameter on CT in mm- median (IQR) | 21.9 (16.5-32.4) | |
| Spiculated | 21 | 44.7 |
| Non spiculated | 26 | 55.3 |
| Solid | 33 | 70.2 |
| Semi-solid | 12 | 25.5 |
| GGO | 2 | 4.3 |
| Bronchus sign present | 29 | 61.7 |
| EMN Samples Results | ||
| Positive EMN guided samples - any type | 31 | 66 |
| Positive EMN TBBX | 26 | 55.3 |
| Positive EMN brushing | 10 | 21.3 |
| Positive EMN TBNA | 4 | 8.5 |
| EMN contributed meaningfully to the diagnosis | 41 | 87.2 |
| EMN with Addition of: | ||
| Radial EBUS | 6 | 12.8 |
| Fluoroscopy | 45 | 95.7 |
| ROSE | 24 | 51.1 |
| No. of Targets Sampled | ||
| 1 | 40 | 85.1 |
| 2 | 7 | 14.9 |
| Lobes Sampled | ||
| RUL | 7 | 14.9 |
| RML | 4 | 8.5 |
| RLL | 15 | 31.9 |
| LUL | 10 | 21.3 |
| LLL | 8 | 17 |
| Lingula | 1 | 2.1 |
| Non-EMN Sampling during the Same Procedure | ||
| Brushing | 8 | |
| BAL | 33 | |
| EBUS FNA | 16 | |
| Total number of patients who had non-EMN sampling during the same procedure | 42 | 89.4 |
| Diagnosis obtained from the non-EMN guided sample during the same procedure | 12 | 25.5 |
| Required Further Invasive Testing | ||
| Yes | 6 | 12.8 |
| CT guided lung biopsy | 4 | |
| VATS/surgical | 1 | |
| CT guided non-thoracic biopsy | 1 | |
Twenty-one of 29 (72%) patients with positive bronchus signs had positive EMNB-guided samples, compared to 10/18 (56%) patients without bronchus signs; however, the difference was not statistically significant (p-value: 0.335). Similarly, a solid vs. non-solid lesion on CT did not result in a significant difference in the positivity of EMNB guided samples (p-value: 0.412). However, when the EMNB sample positivity in patients was compared with malignant vs. non-malignant disease, there was a difference in favor of malignant disease (p-value 0.048). When the presence or absence of ROSE was compared during the procedure, a trend favoring the presence of ROSE could be seen, but this was statistically non-significant (p-value: 0.078).
Table 2 shows a comparison of EMN sample positivity between groups.
The major expected complications due to EMNB are pneumothorax not being related to patient's illness, punctured viscus not being related to patient's illness, bleeding leading to hemodynamic instability or requirement for blood transfusion, unplanned admission or escalation of the level of care, cardiac arrest, cardiac ischemia requiring intervention, dysrhythmia requiring intervention, death and unexpected respiratory failure requiring unplanned intubation or assisted ventilation [1, 2].
In this series, one patient with pre-existing triple vessel coronary artery disease developed an inferior wall ST-segment elevation myocardial infarction (STEMI). He spent one-day post-procedure in the intensive care unit and had a cardiac catheterization. No culprit vessel was identified, and it was felt that the STEMI was likely secondary to a spasm. This patient recovered completely and was discharged from the hospital.
| - | n | EMN Positive | P-Value |
|---|---|---|---|
| Bronchus sign | 29 | 21 (72%) | 0.335 |
| No Bronchus sign | 18 | 10 (55%) | |
| With ROSE | 24 | 18 (75%) | 0. 072 |
| Without ROSE | 23 | 13 (57%) | |
| Solid | 33 | 24 (73%) | 0.412 |
| Non-solid | 14 | 7 (50%) | |
| Malignant diagnosis | 23 | 20 (87%) | 0.048 |
| Non-malignant diagnosis | 24 | 11 (46%) |
4. DISCUSSION
This pilot study demonstrates the feasibility and safety of performing EMNB in our local population. Our group set up the first multidisciplinary interventional pulmonology program in the country, and our initial EMNB sampling results are comparable with those published in large multinational trials. We observed a sample positivity rate of 66%, with only 12.7% of patients requiring additional biopsy procedures.
Becker et al. published one of the first pilot studies on the use of EMNB in 2005. They were able to obtain conclusive biopsies in 20 of 29 (69%) patients, with lesions ranging from 12 to 106 mm in diameter [4]. This was followed by a larger study published by Gildea and colleagues. They used EMNB for peripheral lesions as well as lymph nodes. Their mean peripheral lesion and lymph node sizes were 22.8 (SD12.6) mm and 28.1 (SD 12.8) mm, respectively, and they were able to achieve a diagnosis in 80.4% of the procedures [1].
Since that time, a number of individual studies have been published, showing similar results. Gex and colleagues [5] performed a meta-analysis in 2014 of 15 trials that had been conducted on the use of EMNB and also observed that a definitive diagnosis could be obtained in 64.9% of the cases (1033 lung nodules in 971 patients). Following this, 15 academic centers in the United States published the results of a combined registry called the AQuIRE registry [2]. They noted that bronchoscopy was diagnostic in 312 (53.7%) patients. However, when EMNB alone was used, the diagnostic yield was 38.5%, which increased to 47.1% when EMNB was combined with radial EBUS (r-EBUS). In this “real-world study,” they found that performing peripheral transbronchial needle aspirate or TBNA (as opposed to just transbronchial lung biopsies) improved diagnostic yield. Other factors associated with a higher yield included larger lesion size, non-upper lobe location, and tobacco use. More recently, Folch et al. [6] published the results of the NAVIGATE study, which was a prospective multicenter cohort study enrolling 1215 consecutive patients from 29 sites in the United States. Their median lesion size of 20.0 mm was similar to ours of 21.9 mm. They observed a 12-month diagnostic yield of 73%, with 44.3% of cases having a diagnosis of malignancy. Significant predictors of a diagnostic procedure on the multivariate analysis included a personal history of cancer, use of three sampling tools, lymph node sampling during the EMN procedure, presence of a bronchus sign, biopsy of multiple lesions, and procedure time of less than 60 minutes. They observed no significant difference in yield when radial-EBUS was used. Among the 80 subjects in the study who had a diagnosis of adenocarcinoma and in whom molecular testing was attempted, the investigators reported adequate tissue in 86.2% of the cases.
The results of our study are in line with those published in the most recent NAVIGATE trial [6] and the above-mentioned meta-analysis by Gex et al. [5]. In our experience, not only was EMNB safe, with only one complication reported in our study but it also highly likely prevented the need for more invasive studies with only one patient requiring VATS surgical biopsy. This patient’s final diagnosis after VATS biopsy was combined with small cell carcinoma. The other five patients who needed further diagnostic procedures had CT-guided biopsies, with the final diagnosis being infection in three cases, a rheumatoid nodule in one case, and metastatic malignancy in one case. Of these six patients, only one had a bronchus sign on a CT scan, which may help explain the negative result on EMNB.
One well-documented limitation of EMNB is the low negative predictive value of the test. In this series, of the 16 negative EMNB patients, six went for additional testing, as detailed in the paragraph above. The remaining ten patients’ files were reviewed. Two were lost to follow-up, four were true negatives at 12 months follow up, while the remaining four were deemed to be false negatives, with one patient subsequently diagnosed with metastatic malignancy, two with vasculitis, and one patient had a non-tuberculous mycobacterial infection.
The major limitations of this study are its retrospective design and limited samples size. Since this was a pilot study designed to show the safety and efficacy of the procedure in our specific population, we feel that in the future, larger prospective cohort studies involving multiple sites will be needed to demonstrate if this technology can be effectively used in multiple medical centers rather than being confined to a few quaternary referral centers.
CONCLUSION
In conclusion, this study demonstrates that EMNB can be safely performed in a Middle Eastern population with results similar to those reported in major international studies. The highest diagnostic yield was in patients with a bronchus sign on a CT scan, and combining EMNB with ROSE can increase the chances of having a positive diagnostic bronchoscopy. However, patients and physicians need to be aware of the need to follow up with the patients with negative biopsies to ensure that false negatives are not missed.
LIST OF ABBREVIATIONS
| STEMI | = ST-Segment Elevation Myocardial Infarction |
| EMNB | = Electromagnetic Navigation Bronchoscopy |
| GPS | = Global Positioning Satellite |
| CT | = Computerized Tomography |
ETHICS APPROVAL AND CONSENT TO PARTICIPATE
The study was approved by the Cleveland Clinic Abu Dhabi Research Ethics Committee (REC) (REC Number: A-2019-061).
HUMAN AND ANIMAL RIGHTS
No animals were used in this study. The study on humans was conducted in accordance with the principles of the Declaration of Helsinki (http://www.wma.net/en/30publi cations/10policies/b3/index.html).
CONSENT FOR PUBLICATION
Study participants gave written informed consent. Since this was a retrospective observational study and all data was anonymized, the Clinic Abu Dhabi Research Ethics Committee waived the need for the same.
STANDARDS OF REPORTING
STROBE guidelines were followed.
AVAILABILITY OF DATA AND MATERIALS
The data will be available on the request from the corresponding authors [M.U].
FUNDING
None.
CONFLICT OF INTEREST
Dr. Ali Saeed Wahla is the EABM of the journal The Open Respiratory Medicine Journal. The authors declare no conflict of interest, financial or otherwise.
ACKNOWLEDGEMENTS
Declared none.


