All published articles of this journal are available on ScienceDirect.
Assessment of Inter-relationship between Anemia and COPD In Accordance with Altitude
Abstract
Background:
Anemia, a common blood disorder has been analysed for the risk for chronic obstructive pulmonary disease (COPD), a predominant respiratory condition. The COPD patients have been found to have anemia due to inflammation, accounting for about 7.5-33%.
Objectives:
To predict the influence of anemia in the COPD occurrence as well as to depict the role of change in altitudes in the occurrence of anemic as well as COPD.
Methodology:
A total of 17 hematological parameters has been analysed in the anemic and COPD patients along with the control samples of low and high altitude regions and the significance of the results has been calculated.
Results:
The hematological parameter comparison between anemia and COPD samples of two different altitudes showed highly significant variations (p<0.001) as well as slighter significant variations (p<0.01, p<0.05 and p<0.1) in most of the parameters as well as the prevalence of anemic condition among the COPD patients was significant.
Conclusion:
The considered hematological parameters shown to have a considerable influence in the occurrence of anemia and COPD as well as in the occurrence of anemia in COPD patients along with the change in altitudes.
1. INTRODUCTION
Anemia is a predominant disorder due to the absence of sufficient hemoglobin concentration in whole blood [1, 2]. The prevalence of anemia is highest among the world's population, which accounts for about 24.8%, i.e., approximately 1.62 billion people were accounted to be anemic by the World Health Organisation [3]. The anemic condition is influenced by various factors like age, sex, lifestyle, and altitudinal ranges [4]. The most widespread reason for the anemic condition is a nutritional deficiency, which is observed most predominantly in developing countries worldwide [4, 5].
Chronic obstructive pulmonary disease (COPD) is a respiratory condition characterised by the limitation of airflow in the lungs, which has been considered the third leading cause of mortality [6].COPD accounts for about 10.1% of prevalence and 5.7% of mortality worldwide [7]. Co-morbidities in COPD worsen the condition rapidly, and anemia has also been found to be a co-morbidity in COPD patients. Approximately 86 to 98% of COPD patients are found to have co-morbidities that can lead to the severity of the disease [8-11]. COPD patients accounting for about 20 to 30% in total, approximately, have been observed to have anemia, which is either normocytic or normochromic in nature [12, 13].
The white blood cell (WBC) parameters (WBC count, lymphocytes, neutrophil, mixed cell count (MxD), red blood cell (RBC) parameters (RBC count, hemoglobin, hematocrit, mean corpuscular volume (MCV), mean corpuscular hemoglobin (MCH), mean corpuscular hemoglobin concentration (MCHC), red cell distribution width – standard deviation and coefficient variation (RDW-SD and CV)) and the platelet parameters (platelet count, mean platelet volume (MPV), plateletcrit (PCT), platelet distribution width (PDW), platelet – large cell ratio (P-LCR) are the major hematological parameters and among these, RBC parameters are mainly studied in the diagnosis of the anemic condition.
Although the anemic condition is being found to be influenced by many environmental factors, altitude may have a great impact [14]. The studies have suggested that the forced vital capacity (FVC) measurement of the individuals in the elevated altitudinal ranges showed reduced functional activity, which indicates the role of altitude increase in lung function [15]. Hence, the current study has been framed to test the hypothesis that altitude plays an important role in anemia as well as COPD. The study has also aimed at analysing the interrelationship between anemia and COPD based on the biochemical parameters since the prevalence of anemia in COPD patients has been found to be 7.5% to 33% [16, 17].
2. 2.MATERIALS AND METHODS
2.1. Sample Collection
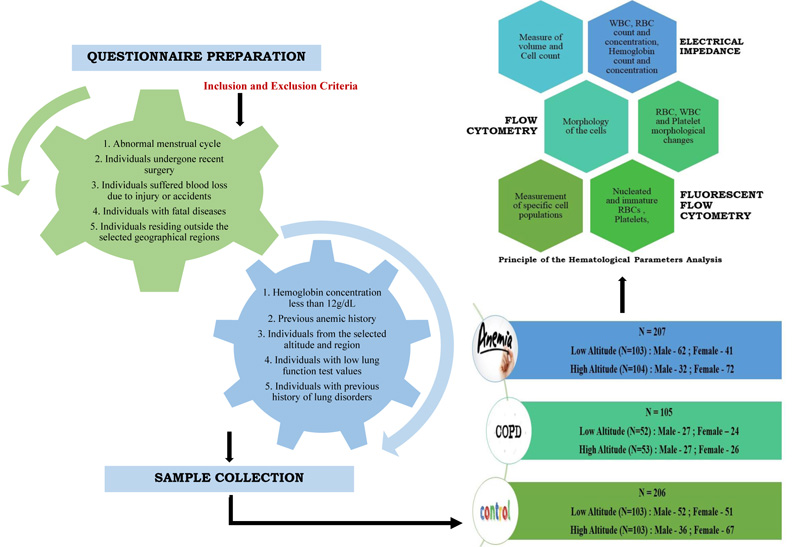
The blood samples were collected from the anemic patients (hemoglobin content, less than 12g/dL), COPD affected individuals (based on a lung function report), and the control individuals (devoid of clinical evidence of anemia and COPD) after obtaining the questionnaire followed by the proper consent form. The Human Ethical Clearance certificate has been obtained from the Avinashilingam Institute for Home Science and Higher Education for Women (Approval No.: AUW/IHEC–18–19/HGMB/FHP–21) for collecting the blood sample from the individuals. The sample collection was done irrespective of age, gender, and race. The sample collection areas include in and around Coimbatore (altitudinal range – approximately 411m above sea level) and in and around The Nilgiris (altitudinal range – approximately 2240m above the sea level). The samples obtained from the Coimbatore region have been categorised as low altitude region samples and The Nilgiris samples have been categorised as high altitude region samples. About 518 individuals were selected based on the questionnaire, the samples were collected in the EDTA tubes, and the analysis was carried out within 24 hours of sample collection. The number of samples collected is given in (Table 1).
2.2. Hematological Analysis
The hematological parameters can be considered the major indicators of clinical conditions of blood [18]. The present study has focused on the analysis of 17 blood parameters, which includes hemoglobin, RBC, hematocrit, MCV, MCH, MCHC, RDW-SD, RDW-CV, WBC, platelets, lymphocytes, MxD, neutrophil, PDW, MPV, P-LCR, and plateletcrit in the obtained blood samples of anemia and COPD patients from the low and high altitude regions. The hematological parameters of the collected samples (n=518) have been carried out in the automated hematological analyser (Mindray, BC 2800) in sterile laboratory conditions. The level of the hematological parameters from the whole blood is determined by two different principles such as electrical impedance, which measures the cell volume or cell count, and flow cytometry, which differentiates the cells by morphological changes.
| Name of the sample | Low Altitude (n) | High Altitude (n) | Total (n) | ||
| Male | Female | Male | Female | ||
| Anemia | 62 | 41 | 32 | 72 | 207 |
| COPD | 27 | 24 | 27 | 26 | 105 |
| Control | 52 | 51 | 36 | 67 | 206 |
2.3. Statistical Analysis
The obtained values of the hematological parameters have been then subjected to statistical analysis, one way ANOVA by using the ‘IBM SPSS Statistics for Windows, version 26 (IBM Corp., Armonk, N.Y., USA)'. The significance has been calculated at the typical levels of “p” at 0.001, 0.01, 0.05 and 0.1.
The outline of the current work has been provided in the diagrammatic representation in (Fig. 1).
3. RESULTS
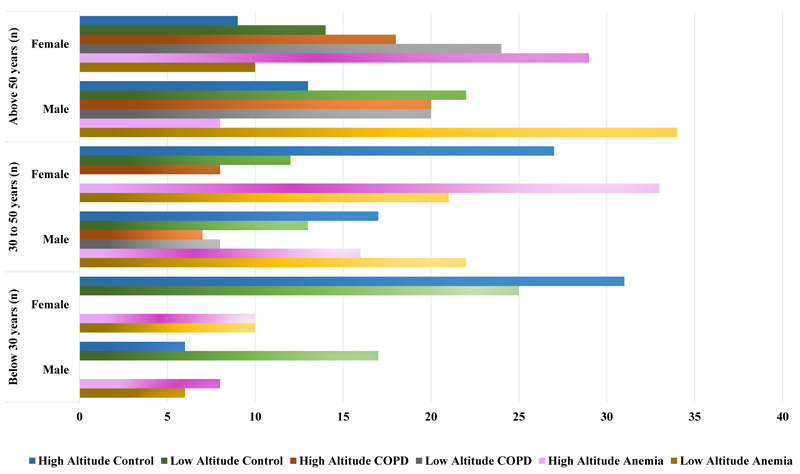
The results of anemia, COPD, and control samples have been obtained in a computerized manner and then tabulated manually for further analysis. The graph (Fig. 2) represents the number of samples and categorisation based on gender and age factors, respectively.
3.1. Altitude Based Comparison
The sample collection has been carried out in two different altitudinal ranges, as mentioned in the methodology section, and the comparison between the same diseases from two different altitudes has been carried out based on the results of the haematological parameters. The anemic patients of the low altitude regions have been compared with the high altitude anemic patients for the prediction of altitude influence on anemia and the same has been followed for COPD and control individuals to predict the altitude influence on the haematological parameters.
The anemic patients were categorised into severe, moderate, and mild anemia based on the hemoglobin content. The hemoglobin range below 8 g/dL was categorised as severely anemic, 8.1 to 10 g/dL as moderate, and 10.1 to 12 g/dL as mild anemia. Out of 103 anemia samples from low altitude regions, 67.96% of patients were severely anemic, 17.47% had moderate anemia, and 14.56% had mild anemia. In the case of 104 high altitude anemic patients, 24.03% were severely anemic, 41.30% were moderately affected, and 34.60% had mild anemia. The 17 haematological parameters considered for the analysis have been tabulated and the statistical analysis has been performed for the prediction of significance.
A highly significant variation (p<0.001) has been obtained in the hemoglobin count, MCH, MCHC, RDW-SD, RDW-CV, WBC range, lymphocytes, MxD, platelet, PDW, and plateletcrit values of the anemic patients of low altitude and high altitude, whereas P-LCR values showed slightly higher significant variation (p<0.01). The anemic patients in comparison with control samples of the respective altitude, showed highly significant variation (p<0.001) in RBC, hemoglobin, and hematocrit values and the low altitude anemic samples showed highly significant variation (p<0.001) in MxD and P-LCR values, whereas RDW-CV values showed slightly higher significance (p<0.01) and the lymphocytes showed significant variation (p<0.05). The MCH and MCHC values showed higher significance (p<0.001) in comparing the high altitude anemic with high altitude control and low altitude anemic samples.
About 84.6% of COPD patients from low altitudes and 79.2% of COPD patients from high altitudes were found to be anemic. The COPD samples of low altitude region in comparison with the respective control samples, showed highly significant variation (p<0.001) in terms of RBC, hemoglobin, hematocrit, and RDW-CV, whereas significant variance (p<0.01) in RDW-SD values and slighter significance (p<0.05) in terms of P-LCR values. The comparison between the COPD and control samples of high altitude regions revealed a higher significance (p<0.001) in RBC, hemoglobin, hematocrit, and neutrophil values and the rest of the parameters were not statistically significant. Among the 17 parameters analysed, only the WBC and neutrophil count of the COPD patients showed significant variation (p<0.001) in comparison between the COPD patients of two different altitudinal ranges.
The anemic patient samples have also been compared with the COPD samples of the corresponding altitudes. The COPD patients were categorised into anemic and non-anemic based on the hemoglobin content. Out of 52 COPD patients from the low altitude region, 84.61% were anemic and 15.38% were non-anemic. From the 53 COPD patient samples of high altitude regions, it has been observed that about 79.2% were anemic and 20.7% were non-anemic. The RBC, hemoglobin, hematocrit, and MxD values of COPD and anemic patients of both the low and high altitudes showed higher significant variation (p<0.001), whereas the high altitude anemic and COPD patients showed higher significance (p<0.001) in terms of MCH, RDW-SD, RDW-CV, lymphocytes, neutrophil, and PDW and the slighter significance (p<0.05) range has been observed in platelet alone.
Apart from the comparison of diseased samples of two different altitudes, the control samples of two different altitudes have been compared to depict the role of altitude in the variations in the hematological parameters. The results showed a slighter significant range (p<0.05) in RBC values and higher significant value (p<0.01) in RDW-CV and a very higher significant variation (p<0.001) in platelet values. The p values obtained for all the hematological parameters of the anemic, COPD, and control individuals from two different altitudes have been tabulated and given in (Tables 2 and 3) shows the information on the ranges of hematological parameters along with the number of individuals falling under each category based on low, normal, and higher range of parameter values and gender.
Table 2.
| AL Vs AH | AL Vs CL | AL Vs CONL | AH Vs CH | AH Vs CONH | CL Vs CH | CL Vs CONL | CH Vs CONH | CONL Vs CONH | |
| RBC | NS* | p<0.001 | p<0.001 | p<0.001 | p<0.001 | NS* | p<0.001 | p<0.001 | p<0.05 |
| Hemoglobin | p<0.001 | p<0.001 | p<0.001 | p<0.001 | p<0.001 | NS* | p<0.001 | p<0.001 | NS* |
| Hematocrit | NS* | p<0.001 | p<0.001 | p<0.001 | p<0.001 | NS* | p<0.001 | p<0.001 | NS* |
| MCV | NS* | NS* | NS* | NS* | NS* | NS* | NS* | NS* | NS* |
| MCH | p<0.001 | NS* | NS* | p<0.001 | p<0.001 | NS* | NS* | NS* | NS* |
| MCHC | p<0.001 | NS* | NS* | NS* | p<0.001 | NS* | NS* | NS* | NS* |
| RDW-SD | p<0.001 | NS* | NS* | p<0.001 | NS* | NS* | p<0.001 | NS* | NS* |
| RDW-CV | p<0.001 | NS* | p<0.001 | p<0.001 | p<0.001 | NS* | p<0.001 | NS* | p<0.01 |
| WBC | p<0.001 | NS* | NS* | NS* | p<0.001 | p<0.001 | NS* | NS* | NS* |
| Lymphocytes | p<0.001 | NS* | p<0.05 | p<0.001 | p<0.001 | NS* | NS* | NS* | NS* |
| MxD | p<0.001 | p<0.001 | p<0.001 | p<0.001 | p<0.001 | NS* | NS* | NS* | NS* |
| Neutrophil | NS* | NS* | NS* | p<0.001 | NS* | p<0.001 | NS* | p<0.001 | NS* |
| Platelet | p<0.001 | NS* | NS* | p<0.05 | p<0.001 | NS* | NS* | NS* | NS* |
| PDW | p<0.001 | NS* | NS* | p<0.001 | NS* | NS* | NS* | NS* | p<0.001 |
| MPV | NS* | NS* | NS* | NS* | NS* | NS* | NS* | NS* | NS* |
| P-LCR | p<0.001 | NS* | p<0.001 | NS* | NS* | NS* | p<0.05 | NS* | NS* |
| PCT | p<0.001 | NS* | NS* | NS* | p<0.001 | NS* | NS* | NS* | NS* |
| Parameters | Anemia | COPD | Control | |||||||||||||||||||||||||||||||||
| Low Altitude | High Altitude | Low Altitude | High Altitude | Low Altitude | High Altitude | |||||||||||||||||||||||||||||||
| ↓ | ↔ | ↑ | ↓ | ↔ | ↑ | ↓ | ↔ | ↑ | ↓ | ↔ | ↑ | ↓ | ↔ | ↑ | ↓ | ↔ | ↑ | |||||||||||||||||||
| M | F | M | F | M | F | M | F | M | F | M | F | M | F | M | F | M | F | M | F | M | F | M | F | M | F | M | F | M | F | M | F | M | F | M | F | |
| RBC PARAMETERS | ||||||||||||||||||||||||||||||||||||
| RBC | 60 | 37 | 02 | 04 | 0 | 0 | 31 | 61 | 0 | 11 | 01 | 0 | 23 | 20 | 05 | 02 | 0 | 02 | 23 | 19 | 04 | 07 | 0 | 0 | 21 | 5 | 31 | 46 | 0 | 0 | 09 | 01 | 22 | 59 | 05 | 07 |
| Hemoglobin | 62 | 41 | 0 | 0 | 0 | 0 | 32 | 72 | 0 | 0 | 0 | 0 | 23 | 21 | 05 | 03 | 0 | 0 | 25 | 19 | 02 | 07 | 0 | 0 | 0 | 0 | 52 | 51 | 0 | 0 | 0 | 0 | 36 | 67 | 0 | 0 |
| Hematocrit | 60 | 41 | 01 | 0 | 01 | 0 | 31 | 70 | 01 | 02 | 0 | 0 | 22 | 20 | 06 | 04 | 0 | 0 | 24 | 16 | 03 | 10 | 0 | 0 | 13 | 12 | 39 | 39 | 0 | 0 | 07 | 06 | 22 | 60 | 07 | 01 |
| MCV | 30 | 09 | 22 | 29 | 10 | 03 | 18 | 52 | 0 | 0 | 14 | 20 | 04 | 04 | 21 | 19 | 03 | 01 | 04 | 06 | 18 | 19 | 05 | 01 | 04 | 13 | 48 | 37 | 0 | 01 | 04 | 08 | 29 | 59 | 03 | 0 |
| MCH | 15 | 18 | 32 | 21 | 15 | 02 | 05 | 24 | 08 | 15 | 19 | 33 | 06 | 06 | 16 | 14 | 06 | 04 | 09 | 08 | 14 | 14 | 04 | 04 | 04 | 15 | 42 | 30 | 06 | 06 | 11 | 13 | 23 | 52 | 02 | 02 |
| MCHC | 15 | 14 | 43 | 26 | 04 | 01 | 09 | 15 | 14 | 35 | 09 | 22 | 03 | 04 | 25 | 20 | 0 | 0 | 06 | 02 | 21 | 22 | 0 | 02 | 02 | 06 | 49 | 44 | 01 | 01 | 04 | 09 | 31 | 57 | 01 | 01 |
| RDW-SD | 02 | 02 | 27 | 16 | 33 | 23 | 02 | 0 | 0 | 02 | 30 | 70 | 0 | 0 | 09 | 08 | 19 | 16 | 0 | 0 | 12 | 10 | 15 | 16 | 0 | 01 | 50 | 44 | 02 | 06 | 03 | 03 | 23 | 50 | 10 | 14 |
| RDW-CV | 02 | 01 | 60 | 40 | 0 | 0 | 0 | 0 | 32 | 72 | 0 | 0 | 0 | 0 | 28 | 24 | 0 | 0 | 0 | 0 | 27 | 26 | 0 | 0 | 10 | 08 | 42 | 43 | 0 | 0 | 0 | 01 | 36 | 67 | 0 | 0 |
| WBC PARAMETERS | ||||||||||||||||||||||||||||||||||||
| WBC | 08 | 06 | 42 | 24 | 12 | 11 | 03 | 07 | 16 | 23 | 13 | 42 | 03 | 04 | 20 | 15 | 05 | 05 | 0 | 0 | 12 | 17 | 15 | 09 | 01 | 0 | 50 | 47 | 01 | 04 | 01 | 02 | 33 | 63 | 02 | 02 |
| Lymphocytes | 10 | 04 | 52 | 36 | 0 | 01 | 0 | 0 | 23 | 39 | 09 | 33 | 09 | 06 | 19 | 17 | 0 | 01 | 03 | 02 | 19 | 25 | 05 | 0 | 0 | 0 | 52 | 51 | 0 | 0 | 0 | 0 | 36 | 67 | 0 | 0 |
| MxD | 10 | 04 | 18 | 13 | 34 | 24 | 01 | 02 | 01 | 14 | 30 | 56 | 11 | 07 | 11 | 12 | 06 | 05 | 13 | 11 | 07 | 08 | 07 | 07 | 0 | 0 | 49 | 49 | 03 | 02 | 0 | 0 | 34 | 67 | 02 | 0 |
| Neutrophil | 0 | 0 | 52 | 33 | 10 | 08 | 04 | 07 | 24 | 54 | 04 | 11 | 0 | 0 | 27 | 24 | 01 | 0 | 0 | 0 | 17 | 16 | 10 | 10 | 02 | 04 | 45 | 43 | 05 | 04 | 13 | 02 | 21 | 63 | 02 | 02 |
| PLATELET PARAMETERS | ||||||||||||||||||||||||||||||||||||
| Platelet | 11 | 09 | 49 | 30 | 02 | 02 | 05 | 16 | 23 | 42 | 04 | 14 | 02 | 01 | 26 | 22 | 0 | 01 | 03 | 02 | 22 | 22 | 02 | 02 | 0 | 0 | 51 | 51 | 01 | 0 | 02 | 02 | 34 | 65 | 0 | 0 |
| PDW | 02 | 01 | 60 | 40 | 0 | 0 | 0 | 0 | 32 | 72 | 0 | 0 | 0 | 0 | 28 | 24 | 0 | 0 | 0 | 0 | 27 | 26 | 0 | 0 | 01 | 0 | 37 | 38 | 14 | 13 | 0 | 02 | 13 | 34 | 23 | 31 |
| MPV | 30 | 20 | 32 | 21 | 0 | 0 | 17 | 44 | 05 | 13 | 10 | 15 | 17 | 15 | 06 | 06 | 05 | 03 | 15 | 14 | 06 | 08 | 06 | 04 | 14 | 17 | 35 | 31 | 03 | 03 | 17 | 21 | 19 | 45 | 0 | 01 |
| P-LCR | 01 | 01 | 61 | 40 | 0 | 0 | 0 | 0 | 32 | 72 | 30 | 0 | 01 | 06 | 27 | 18 | 0 | 0 | 08 | 03 | 19 | 24 | 0 | 0 | 01 | 01 | 51 | 51 | 0 | 0 | 0 | 02 | 36 | 65 | 0 | 0 |
| Plateletcrit | 11 | 03 | 51 | 38 | 0 | 0 | 09 | 28 | 17 | 29 | 17 | 15 | 04 | 02 | 24 | 21 | 0 | 01 | 0 | 0 | 27 | 25 | 0 | 01 | 0 | 02 | 51 | 49 | 01 | 0 | 05 | 07 | 31 | 59 | 0 | 01 |
4. DISCUSSION
The predominant function of the hemoglobin pigment present in RBCs is to deliver the oxygen molecules to the tissues and organs throughout the human body [19]. The level of air circulation decreases with the increase in altitude [20], which in turn reduces the oxygen content inhaled by the human body and delivered to the organs and tissues. The lack of oxygen delivery to the tissues and organs results in a condition called hypoxia, which results in anemia. Studies have revealed that hypoxia may be caused due to higher altitudes, which supports our hypothesis that an increase in altitude influences anemic conditions [20, 21]. The current research has concluded that hematological parameters are influenced to a certain extent in the anemic patients in case of an increase in altitudinal ranges. The variation among the different hematological parameters based on the calculated standard deviation has been given in the graphical representation (Fig. 3).
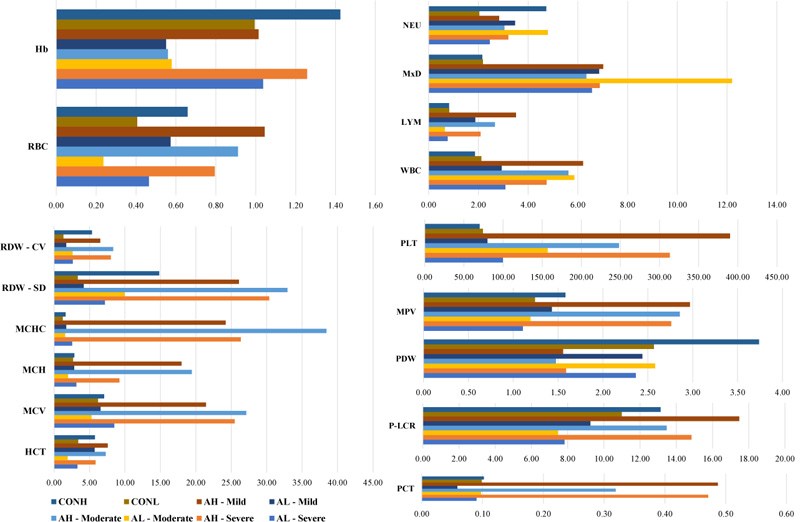
On examining these hematological parameters of the COPD patients, the previous study has suggested that hemoglobin and RBC levels were declined, but the MCH, MCV, and MCHC values were unaltered along with the rise in RDW ranges [22]. The current research on the hematological analysis of COPD patients has also revealed similar results, in which other than RDW-SD, all the RBC parameters such as RBC count, hemoglobin, hematocrit, MCV, MCH, and MCHC showed the values around the normal ranges indicating the absence of abnormality. The hematocrit values have been observed to be declined in anemic conditions, whereas contrastingly, the levels were increased in COPD patients, although the interlink between COPD and hematocrit remains unclear [23-25]. Contrastingly, the hematocrit values of the COPD patients were comparatively around normal ranges in the present research work.
The MCV levels are altered in both anemic and COPD conditions [26, 27]. The presence of anemia in the COPD condition has various etiological sources, including the high-level synthesis of cytokines that leads to the reduction in synthesis of erythropoietin or improper iron absorption as a result of insufficient hepcidin, a hormone that is responsible for the iron metabolism regulation [28]. In the current research involving COPD and anemic patients, the anemic condition among the considered COPD patients was around 79.20%, which strongly indicates the prevalence of anemia among COPD patients.
The immune cells, which are the WBCs of the human body, have no significant association with anemia but have a significant association with COPD condition [29, 30]. Although the WBC parameters of anemic patients did not show significant variations, the COPD individuals exhibited significant changes in immune response cells in the current research work. Apart from RBC and WBC parameters, platelet parameters have been considered in the present study. But, the previous studies have suggested that platelet stability has been linked with the stage of severity of anemia [31, 32]. COPD patients have been found to associate with elevated levels of platelet parameters, which may be caused by inflammatory conditions in the respiratory system [32]. Contrastingly, the present study did not reveal any significant changes in the platelet parameters of the patients.
CONCLUSION
Anemic condition is not considered fatal when compared with the COPD condition. But, the combination of both anemia and COPD has been expected to decrease the life expectancy considerably; hence, the current study has been aimed at depicting the influence of hematological parameters and altitude in comparison with both diseases. From all the above observations, it can be concluded that anemic condition is significantly influenced by the increase in altitude ranges and the influence of anemic condition on COPD is considerably higher.
LIMITATIONS OF THE STUDY
The current study involves the two different altitudinal ranges but the sample collection does not involve gender categorisation. The numbers of samples collected from males and females are unequal, which makes the current study has to be extended with an equal number of samples to support the obtained results.
LIST OF ABBREVIATIONS
| COPD | = Chronic Obstructive Pulmonary Disease |
| RBC | = Red Blood Cells |
| MCV | = Mean Corpuscular Volume |
| MCH | = Mean Corpuscular Hemoglobin |
| MCHC | = Mean Corpuscular Hemoglobin Concentration |
| RDW–SD | = Red Cell Distribution width – Standard Deviation |
| RDW–CV | = Red Cell Distribution width – Coefficient of Variation |
| WBC | = White Blood Cells; MxD- Mast Cell Ratio |
| PDW | = Platelet Distribution width |
| MPV | = Platelet Distribution width |
| PDW | = Mean Platelet Volume |
| P–LCR | = Platelet Large Cell Ratio |
| SD | = Standard Deviation |
| SE | = Standard Error |
AUTHORS' CONTRIBUTIONS
Sangeetha Thangavelu – Sample Collection, Parameter Analysis, Data Collection, Manuscript Preparation; Vijaya Anand Arumugam – Screening of data collection, Manuscript Preparation, and Structuring; Nargis Begum Tajuddin – Reviewing of Manuscript.
ETHICS APPROVAL AND CONSENT TO PARTICIPATE
The Human Ethical Clearance certificate has been obtained from the Avinashilingam Institute for Home Science and Higher Education for Women (Approval No.: AUW/IHEC–18–19/HGMB/FHP–21)
HUMAN AND ANIMAL RIGHTS
No animals were used in this study. The reported experiments on humans are in accordance with the Helsinki Declaration.
CONSENT FOR PUBLICATION
Consent was obtainedby the proper consent form.
AVAILABILITY OF DATA AND MATERIAL
The authors confirm that the data supporting the findings of this study are available within the article.
FUNDING
The study has not been supported by any funding resources.
CONFLICTS OF INTEREST
The authors declare that they have no conflicts of interest.
ACKNOWLEDGEMENTS
Declared none.


