Expression of ΔF508 Cystic Fibrosis Transmembrane Regulator (CFTR) Decreases Membrane Sialylation
Abstract
Chronic colonization and infection of the lung with Pseudomonas aeruginosa is a major cause of morbidity and mortality in cystic fibrosis (CF) patients. Imundo, et al. determined that CF cells had a higher concentration of an asialoganglioside (asialo-GM1), to which both P. aeruginosa and S. aureus bound preferentially. We sought to determine if the expression of mutant CFTR is associated with altered sialylation. Our study of epithelial cells transfected with normal and mutant ΔF508 CFTR, the defect in the majority of CF patients in the United States, were analyzed by ELISA and FACS analysis of cell membranes labeled with lectins which bind to Neu5Ac. We determined that ΔF508 CFTR is associated with decreased membrane sialic acid residues in the α2, 3 position and increased concentrations of asialo- GM1. Quantitation of sialic acids released from the cellular membranes demonstrated that the presence of the ΔF508 CFTR is associated with markedly decreased membrane sialylation, but similar cytoplasmic sialylation. Thus, ΔF508 defect is correlated with decreased expression of GM1 and with decreased sialylation of all cell surface structures, and this change occurs during post-translational modification of glycoproteins and glycolipids. This may be one factor involved in the chronic bacterial colonization seen in these patients.
INTRODUCTION
Cystic fibrosis (CF) is characterized by abnormal epithelial ion and fluid transport and is associated with mutations in the CF gene product, the cystic fibrosis transmembrane conductance regulator (CFTR) protein. Although much about function and regulation of normal CFTR is known [1-3], the precise cellular pathways that are affected in CF are still under investigation. The most common mutation of CFTR, affecting approximately 90% of patients, is a deletion of phenylalanine at codon 508 (ΔF508 CFTR) [2]. The major cause of morbidity in the clinical disease is chronic pulmonary colonization and infection with P. aeruginosa [4-6] and S. aureus. Imundo, et al. [1] has demonstrated that CF epithelial cells have increased binding of P. aeruginosa and S. aureus. Since these bacteria recognize and bind to the GaINAcβ1-4Gal disaccharide sequence in asialo-GM1 and asialo-GM2 [7, 8] preferentially to GM1 and GM2, bacterial colonization may be related to altered sialylation of CF cells. Further work is needed in the study the relationship between sialylation and bacterial colonization in human CF cells. Abnormalities in sialylation of cells transfected with mutant CFTR and secretions of patients with CF have been described [9]; however, it has not been clear whether this phenomenon has been directly mediated by the presence of neuraminidases in colonizing bacteria or by the abnormal CFTR.
Mammalian cells expressing ΔF508 CFTR have been shown to have ion permeability changes, presumably due to the fact that the mutant protein fails to exit from the endoplasmic reticulum (ER) and localize normally in the plasma membrane [10]. The ΔF508 CFTR has normal chloride channel function in the ER [11], and when it reaches the apical membrane in a low temperature environment, as can be performed using cell culture of some cell types. However, under growth conditions at physiologic temperatures, the ΔF508 CFTR has less stability than the normal CFTR [11]. Although cells from CF patients may have abnormal transport pathways, including those driven by agonists (cAMP) [2] and inhibitors ((D)-Ins [3-6] P4) [12, 13], it is not obvious how these differences affect post-translational modification of glycoproteins and glycolipids. Thus the relationship between the Golgi, the cellular compartment in which post-translational modifications such as addition of sialic acids by sialyltransferases occurs [9, 14], and the CFTR may be important in disease pathogenesis.
This study was conducted to qualitatively and quantitatively compare the expression of sialylated glycoconjugates and gangliosides on the cell membranes of CI27 mouse mammary epithelial cells transfected with normal CFTR protein, ΔF508 CFTR, or vector alone, to determine whether altered sialylation is associated with heterologous expression of the CFTR ΔF508 mutation. We used a combination of ELISA, immunocytochemistry and FACS to demonstrate the differential presence of sialic acid residues recognized by specific lectins and quantitative sialic acid analysis, and to determine whether the ΔF508 mutation of the CFTR is responsible for the sialylation defects noted in this common form of cystic fibrosis.
MATERIALS AND METHODS
Reagents
Fluorescein and Biotin conjugates of Maackia amaurensis lectin (MAL II), avidin D-alkaline phosphatase, Vector Laboratories (Burlingame, CA); biotinlylated conjugate of cholera toxin, List Biological Lab, Campbell, CA.; N-acetylneuraminic acid and N-glycolylneuraminic acid1,2 diamino-4,5-methylenedioxybenzene, N-propyl gallate, N-ethylmaleimide, PMSF, leupeptin, TBST, goat anti-rabbit alkaline phosphatase, Sigma Chemical Company, St. Louis, MO; Biorad protein standard assay, alkaline phosphatase substrate kit (p-nitrophenylphosphate, diethanolamine buffer), BioRad, Hercules, CA. All other chemicals were of reagent grade and were purchased from commercial sources.
Transfected Cell Lines
Mouse mammary C127 epithelial cells, which do not express detectable levels of CFTR [15], transfected with bovine papilloma-based vector containing CFTR cDNA (WT), a CFTR cDNA with the ΔF508 mutation (ΔF508) (generously provided by the Genzyme Corporation, Framingham, MA), or vector alone (BPV) were used, as described [15]. These cell lines have been used in previous experiments on CFTR and processing defects and efficiency of transfection was confirmed [15]. Cell lines were maintained in Dulbecco's modified Eagle's medium (DMEM) with 10% fetal calf serum in an atmosphere of 5% CO2 and were passaged each week. Confluent monolayers were used for experiments at days 2-5 after passaging.
Immunocytochemistry
Cells were grown to a monolayer on glass coverslips and fixed with 4% paraformaldehyde in PBS (0.9% NaCI in 10mM sodium phosphate buffer, pH 7.4) containing 5% sucrose for two hours at room temperature; coverslips were then washed with PBS. The cells were incubated with MAL II (20µg/ml) for 60 minutes followed by avidin D-FITC (10µg/ml) for 30 minutes, pH 7.4 at room temperature. The coverslips were washed between incubations with PBS and mounted in PBS-glycerol, pH 8.0, with 2% N-propyl gallate as an anti-quenching agent. The slides were viewed on a Nikkon epifluorescence microscope and photographs were taken on Kodak 3200 film [15].
Membrane Preparation
For ELISA assay, adherent cells were rinsed in cold PBS and then 3-5 ml of homogenization buffer (0.25 M sucrose, 10mM Tris, 1mM EDTA pH 7.4) was added to each dish. The cells were scraped and dounced. The nuclear pellet was discarded after the cells were centrifuged at 500 x g for 10 minutes at 4°C. The supernatant was then centrifuged at 100,000 x g for 2 hours at 4° C. This supernatant (cytosol) and pellet (membrane) were saved at -70° C and a small aliquot from each was used for protein determination by Biorad protein standard assay using BSA as the standard. Membrane protein isolates were treated with lysate buffer (20mM Tris-HCI pH 8.0, 100mM NaCI, 1% Triton X 100, 10mM NEM, 2mM PMSF, 0.1% leupeptin).
For sialic acid analysis adherent cells were rinsed in cold PBS, the plates were scraped and washed three times in ice cold PBS containing 2mM PMSF, 0.1% leupeptin. Pellets resuspended in 500 uL PBS were homogenized using ten 5-second pulses with a Polytron homogenizer (Brinkmann). The nuclear pellet was discarded after the cells were centrifuged at 500 x g for 10 minutes. The supernatant was ultracentifuged at 100,000 x g for 1 hour. The supernatant was reserved and the membrane pellet resuspended in 50 mM Tris, pH 7.4 and a small aliquot saved for protein determination using the method of Lowry [16].
ELISA Assay
High affinity flat bottomed 96 well ELISA plates (Immulon 3, Dynatech Laboratories Inc, Chantilly, Virginia) were coated and incubated with serially diluted membrane proteins, overnight at 4°C. The plates were washed with TBST pH 7.6, Tris buffered saline (IM Tris-HCI, 1M Tris Base, 1.5 M NaCI) with 5% Tween between incubation times. The plates were then blocked with 1% gelatin for one hour. The plates were again washed with TBST. The primary antibody or lectin-biotin was applied to each well. The lectins were diluted in PBS. Cholera Toxin (0.1 mg/ml) conjugated to biotin was used in the same manner as lectins. For assaying binding of lectins and cholera toxin, the wells were then washed and incubated for one hour with avidin D-alkaline phosphatase. The plates were then washed several times and reacted with alkaline phosphatase substrate kit (p-nitrophenyl phosphate, diethanolamine buffer). The reaction was stopped with 2M NaOH and read at 410 nm on an ELISA reader (Dynatech Laboratories Inc. MR700). The p values were calculated by the student t test, or ANOVA as appropriate, using triplicate data points from each of the analyses.
Release and Analysis of Sialic Acids
Sialic acids were released by adding acetic acid to a final of 2M acetic acid, and heating at 80oC for 3 hours [17, 18]. Membranes were pelleted by centrifugation at 12,000 g for 5 minutes; supernatants were centrifuged through a Microcon™ 10,000 m.w. cut off filter for 30 minutes. Supernatants were treated with 0.1 M NaOH for 30 minutes at 22°C followed by addition of an equal volume of 0.1M HCl; they were then kept at -70oC until analysis.
Aliquots containing released sialic acids were derivatized for 2.5 hours at 50oC with DMB, as described [19, 20]. HPLC was carried out using a TSK gel ODS-120T column eluted in the isocratic mode with a mixture of acetonitrile-methanol-water (9:7:84, v/v) at a flow rate of 1.0 ml/min at 22oC. Effluent was monitored using a Linear Fluor LC 304 fluorescence detector at an excitation wavelength 374 emission wavelength 448. Integration areas were compared to sialic acid standards and expressed as picomoles sialic acid per milligram of protein in the sample. Duplicate injections were analyzed from each of four separate membrane preparations.
Flow Cytometry
A fluorescence activated cell sorter (FACScan, Becton Dickinson and Co, Mountain View, CA) was used to determine the presentation of sialo GM1 on the epithelial cell surfaces of WT, ΔF508, and BPV cells using the β-subunit of cholera toxin, which binds to GM1. Confluent layers of cells were dissociated with 0.25% trypsin at 37oC for 10 minutes, washed and resuspended in PBS, pH 7.4. The cells were counted and divided into 1 x105 cell aliquots. The cells were washed in PBS and incubated for 60 minutes at room temperature with cholera toxin β-subunit, diluted 1:50 (0.1 mg/ml) in PBS-BSA. After the primary incubation, cells were washed three times with PBS and reacted with 1:100 Goat anti-rabbit FITC for 60 minutes with end-over-end rotation. After washing in PBS, cells were fixed in 1% paraformaldehyde. Approximately 10,000 events were recorded. An aliquot was used as a control for nonspecific binding using cells from each cell line labeled with FITC conjugate alone.
RESULTS
ΔF508 Cells have Fewer Receptors for Maackia Amaurensis Lectin (MAL II)
MAL II binds to sialic acids in the Neu5Ac α2,3 Gal/GalNAc sequence [21]. As can be seen in Fig. (1), the MAL II binds intensely to the surfaces of WT and BPV cells. Membrane staining is marked by linear fluorescence along the borders of confluent monolayers and diffuse punctate staining over the rest of the cell surfaces. In contrast ΔF508 cells have less staining at both complete confluence and at low density, indicating that plating density is not relevant. This finding suggests that α2, 3 sialic acid moieties that bind to the lectin are present on WT cells but are diminished on the cell surfaces of ΔF508 cells. While this is a qualitative analysis, the differences between staining and absence of staining are evident. The lighter staining observed with the BPV cells, may be artifactual or a reflection of intermediate presentation of sialic acid moieties in cells without CFTR.
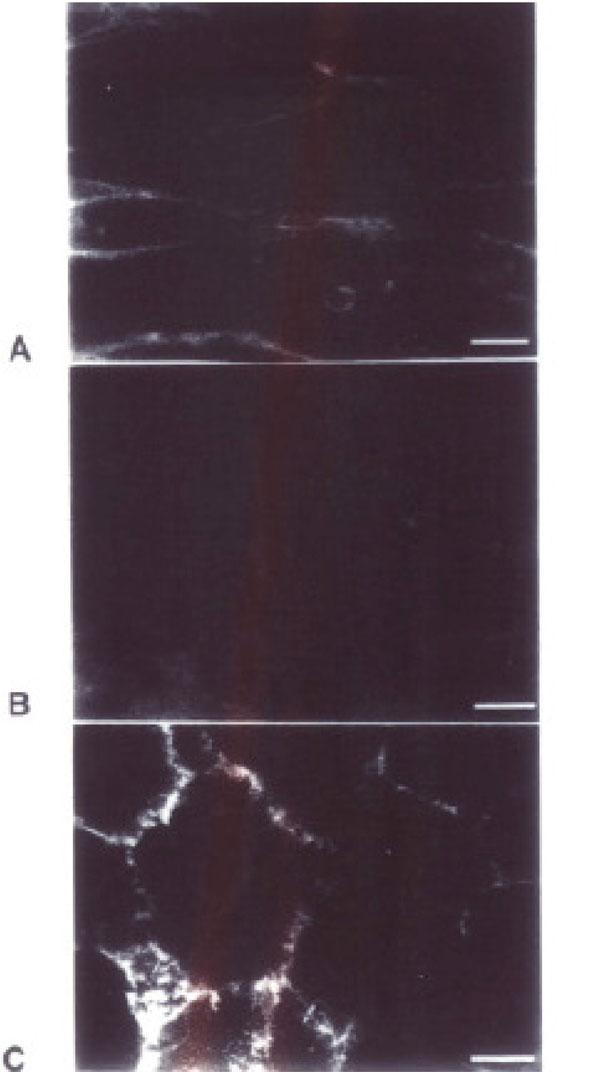
Results of Immunocytochemistry. Differential binding of MAL II Lectin-FITC to transfected cell lines. In panels A (BPV) and C (WT) there is notable binding of MAL II Lectin-FITC, which binds selectively to sialic acid at the α2, 3 position on cell borders. Panel B (ΔF508) has a qualitative decrease in binding to this lectin. Bar =10um.
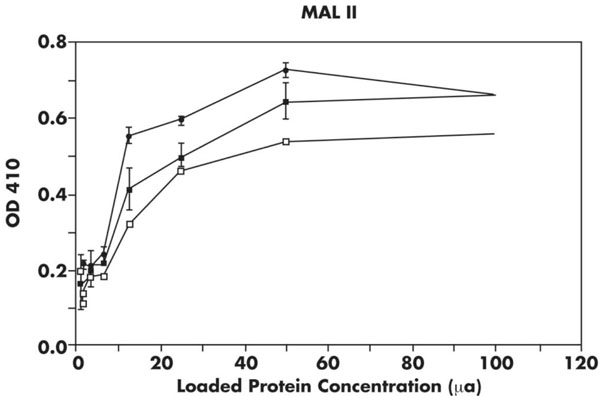
ELISA of cellular membrane isolates using MAL II. Results of incubation with MAL II with several concentrations of membrane proteins by ELISA (see Materials and Methods). Membrane protein concentration is on x-axis and absorbance reading on y-axis. Closed circles, WT; closed squares, BPV; open squares, ΔF508. The ΔF508 membranes have significantly less MAL II binding (p<0.0005) compared to either BPV or WT lines.

ELISA of cellular membrane isolates using cholera toxin β subunit. Cellular membranes from BPV, WT and ΔF508 cell lines were incubated with cholera toxin β subunit, which binds preferentially to GM1. Protein concentration is plotted on the x-axis and absorbance on the y-axis. Closed circles, WT; closed squares depict BPV; ΔF508 open squares. The ΔF508 membranes have decreased binding (p<0.05) compared to WT and BPV cells.
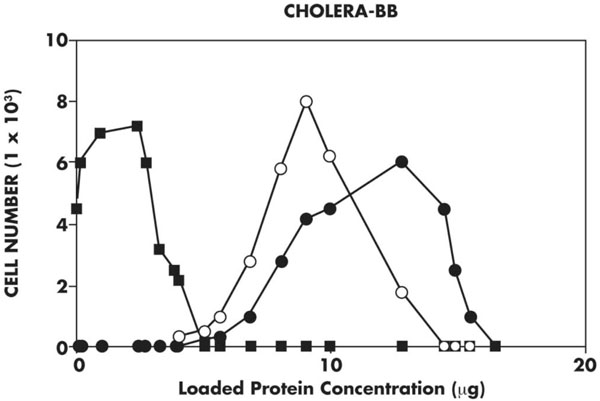
Flow cytometry of cell lines using cholera toxin-biotin probe. FACScan using a cholera toxin-biotin probe reacted with avidin D-Alkaline Phosphatase shows that the ΔF508 cells (closed squares) have less fluorescence than the WT (closed circles) cell lines. This result is consistent with the ELISA shown in Fig. (3).
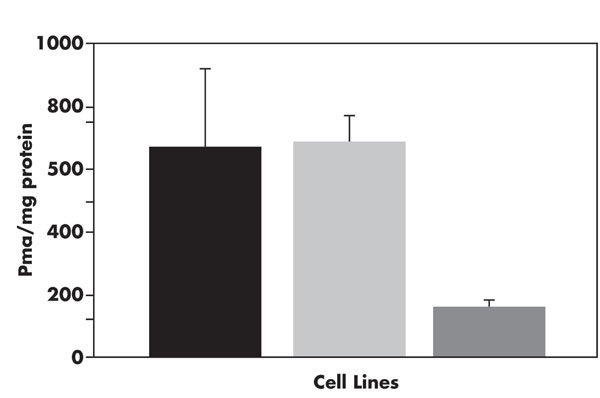
Quantitation of membrane-bound sialic acids. Cells from BPV, WT, and ΔF508 lines were fractionated by ultracentrifugation (see Materials and Methods), sialic acids were released and quantified using derivatization with DMB over a TSK-ODS-120T column. The sialic acids are expressed as picomoles Neu5Ac released relative to protein in the membrane preparation, as determined by the Lowry method. Error calculated using a student t test. The cell lines are designated as: WT, solid; BPV, shaded; ΔF508, clear.
ELISA Assays on Membrane Isolates Demonstrate Differences in Surface Sialic Acids in WT and BPV Cells Compared to ΔF508 Cells
MAL II
As shown in Fig. (2) in which ELISA assays using MAL II are shown, the ΔF508 line have lower absorbance readings at all concentrations ranging from 12.5 µg to 100 µg compared to the WT and BPV cell lines (p<0.0005). The membrane sialic acid α2,3 residues recognized by MAL II of the ΔF508 line are consistently reduced when compared to the WT and BPV cells.
Cholera Toxin
Cholera toxin binds specifically to the carboxyl group of the Neu5Ac moiety and the terminal galactose of GM1 [22]. The binding of cholera toxin β-biotin to the cell membrane isolates was measured by spectrophotometric reaction of Avidin D and substrate with bound cholera toxin β subunit-biotin. The ΔF508 line has significantly less (p<0.05) binding of the cholera toxin β subunit compared to WT and BPV cell lines in nine experiments. The ΔF508 cells have fewer GM1 gangliosides as measured by this assay (Fig. 3). As shown, the absorbance reading that corresponds to the binding of cholera toxin is less in ΔF508 cells compared to either WT or BPV cells.
Flow Cytometry
A FACScan analysis of sialo-GM1 receptors for cholera toxin β-subunit demonstrates that ΔF508 cells have a significantly decreased presentation of such GM1 sites compared to WT cells, as shown in Fig. (4). When cells of each cell line are analyzed by fluorescent emission at 540 nm, there is a consistent shift in fluorescence absorption between ΔF508 and WT cells in two analyses. These results are consistent with those obtained on ELISA.
ΔF508 CFTR cells have decreased surface membrane sialic acids compared to controls.
Total amounts of sialic acids released and purified as described in “Materials and Methods" were determined by comparison of calculated area under the curve of duplicate samples compared to sialic acid standards. As has been previously demonstrated [19, 20] this technique is sensitive to low picomolar amounts of total sialic acids per mg protein and is reproducible. For this analysis all sialic acids were converted to the parent conformation by treatment with 0.1M NaOH prior to analysis; this step was performed to increase reproducibility of results even though the only sialic acid noted in untreated samples was Neu5Ac. As can be seen in Fig. (5), the ΔF508 membranes have markedly less total membrane sialic acid compared to either WT or BPV cell membranes. This is consistent with the results of FACScan, ELISA and immunocytochemistry results which demonstrate that sialic acids in α2,3 linkages are decreased in ΔF508 cells.
ΔF508 CFTR cells have similar cytoplasmic levels of sialic acids compared to controls.
Total amounts of sialic acids from the cytoplasmic compartment from two separate preparations released and purified as described in “Materials and Methods" were deterdetermined by comparison of calculated area under the curve of duplicate samples compared to sialic acid standards. This analysis demonstrates that the levels of sialic acids in the cytoplasm of all cell types are equivalent: WT: 675 pmol/mg, BPV: 623 pmol/mg, ΔF508: 821 pmol/mg (mean values of duplicates from two separate preparations). This implies that the CFTR is not required for sialic acid synthesis and that the ΔF508 mutation does not alter synthesis of sialic acids.
DISCUSSION
This study shows that the heterologous expression of ΔF508 CFTR in cells which do not normally express CFTR results in a quantitative decrease in the sialylation of cell membrane glycoconjugates. The specific binding of lectins to sialic acids in the Neu5Acα2,3 GaIβ1-4GlcNAc linkage is significantly higher in WT and BPV cell lines than in cells with the ÄF508 CFTR. The cells with ΔF508 CFTR also have a lower level of GM1, as evidenced by decreased binding to cholera toxin.
Previous studies have shown an alteration of glycosylation in cystic fibrosis cells and secreted mucous glycoproteins [23]. This study provides direct evidence for a sialylation defect of cells expressing ΔF508 CFTR and corroborates reports using qualitative observations using lectin and antibody binding to detect membrane sialic acids [15].
The precise role of CFTR in the trafficking of proteins and gangliosides to the cell surface has not been elucidated. The results from this study indicate that the CFTR is not required for normal sialylation because the BPV cells and WT cells have equivalent sialylation of cytosolic components. It is possible that the over-expression of the ΔF508 CFTR, retained in the RER at physiologic temperatures, leads to altered sialylation by a previously undescribed pathway. However, it seems more likely that the differences in sialylation are either due to differences in activity of sialyltransferases or to differences in processing and re-utilization of cell membrane components. One hypothesis is that the ΔF508 CFTR defect induces defective acidification of Golgi vesicles, which could decrease activity of pH-dependent sialyltransferases, but Verkman, using ratio-imaging confocal microscopy with liposomal injection into the lumen of trans-Golgi has provided direct evidence that pH of the trans-Golgi compartment is not altered by CFTR expression and activation [24]. One recent study even indicates that hyperacidification of the trans-Golgi network in CF lung epithelial cells leads to undersialylation of plasma membrane glycoconjugates, which in turn promote lung pathology and bacterial colonization [25].
Verkman’s additional studies indicated that the CFTR is involved in endosomal fusion, which could alter processing and distribution of membrane components [3], thus providing an alternative hypothesis for defective post-translational modifications. Duncan and Kornfeld reported that internalized proteins are exposed to a sialyltransferase in the trans-Golgi network in approximately 10% of internalization cycles [14]. Therefore, defective vesicle fusion, defective sialyltransferase activity, or a combination of the two may lead to altered post-translational modification of proteins and lipids, as occurs in the cells expressing ΔF508 CFTR. Metabolic labeling of these cells with sialic acid precursors would determine the temporal sequence for the differences in sialylation, and may help explain the mechanism by which the differences occur.
An increase of asialo-GM1 in cells with the ΔF508 CFTR mutation is important because presentation of the Galβ1-3GalNAcβ1-4 Galβ1-4 Glc-ceramide of asialo-GM1 serves as a receptor for Pseudomonas and other bacteria, such as S. aureus and K. pneumoniae [1]. A decrease in membrane sialylation of GM1 is likely to be responsible for characteristic pulmonary infections that contribute to the morbidity and mortality of CF [26]. It is also likely that altered membrane sialylation plays a role in decreased incidence of gastrointestinal infections with Salmonella typhi [27]. Less clear is whether altered membrane sialylation plays a role in the pathophysiology of chronic pancreatitis, the incidence of which is increased in patients with abnormalities of CFTR [28], but which is apparently unrelated to bacterial infection. Further work will be necessary to clarify whether altered sialylation in cells that express ΔF508 CFTR may contribute to other facets of the clinical presentation. Based on these and other studies the undersialylation of plasma membrane glycoconjugates in patients with CF may promote lung pathology associated with chronic bacterial colonization of the lung.
ABBREVIATIONS
ACKNOWLEDGEMENTS
The authors thank Jennifer Zhou for her much appreciated technical assistance.


